Introduction
The species is the fundamental unit of any study on the biodiversity and ecosystems of our planet, and yet, many groups of organisms have only a small fraction of their species formally described (Gaston Reference Gaston2000). Fungi as a major phylum present in almost all biomes represent a remarkable example: the number of species is expected to be much higher than a million, while only 100 000 are currently described (Blackwell Reference Blackwell2011; Hawksworth Reference Hawksworth2012). We performed detailed anatomical and chemical studies of lichen material recently collected in Réunion (a small and remote island in the Indian Ocean) in a widespread and speciose genus (Micarea Fr., Pilocarpaceae, Lecanorales) to describe each species and formulate a sound taxonomic treatment of all species found in this limited, but very diverse, territory in the tropics. By showing that the genus Micarea is species-rich in Réunion, with many undescribed species, we provide further data for the better assessment of the total number of undescribed lichen taxa (Lücking et al. Reference Lücking, Rivas Plata, Chaves, Umaña and Sipman2009; Bass & Richards Reference Bass and Richards2011).
Although there are many species of Micarea in the Southern Hemisphere, the few monographic treatments of the genus, however masterful, have addressed only European species (Coppins Reference Coppins1983, Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009; Czarnota Reference Czarnota2007). Molecular inferences resolved the genus into different clades and thus it is polyphyletic (Andersen & Ekman Reference Andersen and Ekman2005; Sérusiaux et al. Reference Sérusiaux, Brand, Motiejūnaitė, Orange and Coppins2010; Schmull et al. Reference Schmull, Miądlikowska, Pelzer, Stocker-Wörgötter, Hofstetter, Fraker, Hodkinson, Reeb, Kukwa and Lumbsch2011): M. crassipes is the type species of the distantly related genus Helocarpon, M. sylvicola and its relatives belong to the Psoraceae, and all other species form a supported but poorly resolved clade with all accessions of the Ectolechiaceae and the Pilocarpaceae s. str. nested into it. Further phylogenetic studies with several loci are therefore needed before a well-supported delimitation of the genus can be adopted.
Phylogenetic inferences from mtSSU sequences (Czarnota & Guzow-Krzemińska Reference Czarnota and Guzow-Krzemińska2010) were used to investigate the species delimitation within the well-known, albeit notoriously difficult, Micarea prasina group. Three lineages were resolved within M. micrococca, two being formally recognized at species level [M. byssacea (Th. Fr.) Czarnota et al. and M. micrococca (Körb.) Gams ex Coppins s. str.]. This study assumed that a phylogenetic tree produced with data from a single locus can disentangle a difficult group of species, whereas gene trees represent ‘local optima’ that require additional tests and appropriate statistical methods to yield a robust species tree (Maddison Reference Maddison1997; Edwards Reference Edwards2008; Knowles & Kubatko Reference Knowles, Kubatko, Knowles and Kutbako2010; Camargo et al. Reference Camargo, Avila, Morando and Sites2012a , Reference Camargo, Morando, Avila and Sites b ; Carstens et al. Reference Carstens, Pelletier, Reid and Satler2013). It nevertheless demonstrates that the variation within the M. prasina group is quite substantial and requires detailed investigation.
Even when excluding the species that belong elsewhere in the Lecanorales, the genus is very diverse and occurs on all continents, including the Antarctic, and its diversity is estimated at c. 90 species (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009). The area best sampled and studied is certainly Europe (Czarnota Reference Czarnota2007; Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009). Asia (from the western Himalaya to Japan) and tropical mountains in the three continents represent unexplored and highly promising biomes for studying the diversity of the genus.
Réunion is a small island of 2512 km2 in the Indian Ocean, part of the Mascarene archipelago along with two other islands, Mauritius and Rodrigues. It has a volcanic origin, and one of the most active volcanoes in the world (Piton de la Fournaise) is located on its eastern side, with an elevation of 2631 m. Otherwise it is a serrated mountain range (highest point at 3069 m at the Piton des Neiges), dissected in huge ‘cirques’ and very deep gorges. Its position in the tropical zone makes its climate warm and seasonal, although the temperature can be quite low at high elevations. A working list of lichen species found on the island was published recently (van den Boom et al. Reference van den Boom, Brand, Ertz, Kalb, Magain, Masson, Schiefelbein, Sipman and Sérusiaux2011), based on collections gathered over the last 15 years. A total of 463 taxa were recorded, including several rare species, large expansion of the distribution range for others, and the detection of an austral element (species whose distribution range is limited to the Southern Hemisphere). Finally, analysis of the status of species endemic to the island pointed to the urgent need for taxonomical studies in speciose and poorly known genera. Micarea is one of these and the taxonomy of species found on the island is the aim of this paper.
Materials and Methods
This study is mainly based on extensive collections of Micarea, made by the authors on the island in 2008, and hosted in LG and the private herbaria of the first two authors (h refers to the private herbarium of the collector). Further specimens gathered in 2009 by one of us (ES), with Nicolas Magain, were also included. Anatomical data were gathered and measured in dilute KOH for conidia and paraphyses width, and on material mounted in water for all other characters. In each collection, c. 5 well-developed ascospores representing the size and shape variation detected, conidia and paraphyses were measured up to 0·1 µm accuracy using camera lucida drawings.
Chemical compounds were studied using several methods: medulla under UV light; melting point and microcrystallization; thin-layer chromatography (TLC) using solvent system A (toluene/1,4-dioxane/acetic acid 180:45:5) or C (toluene/acetic acid 170:30) when gyrophoric acid was expected (Huneck & Yoshimura Reference Huneck and Yoshimura1996; Orange et al. Reference Orange, James and White2010) and visualization of spots with sulphuric acid sprayed over the plates, followed by heating at 110°C for c. 5 min. Insoluble pigments are characterized following the classification of Meyer & Printzen (Reference Meyer and Printzen2000).
Results
Chemical compounds
Here we present data on the chemical compounds detected in our material. Details are provided only for unusual compounds or those that can be difficult to distinguish. The numeral in TLC rubric refers to the height, measured in mm, after a 45 mm run; H refers to the spot colour after charring in H2SO4; U2 refers to UV examination at 254 nm, while U3 refers to 360 nm; and finally extract description refers to acetone extraction on a microscope slide.
-
Methoxymicareic acid (Elix et al. Reference Elix, Jones, Lajide, Coppins and James1984)= prasina A (sensu Coppins Reference Coppins1983): TLC 26; U2+, U3± white (“blue-white” in Coppins Reference Coppins1983); H yellow-brown; U3 after charring dark brown; crystals not melting on heating (or only after prolonged boiling).
-
Micareic acid (Elix et al. Reference Elix, Jones, Lajide, Coppins and James1984)=prasina B (sensu Coppins Reference Coppins1983): TLC 26; U2+, U3± white (“blue” but less than prasina A in Coppins Reference Coppins1983); H (orange-) yellow; U3 after charring vivid yellow; crystals melting on gentle heating.
-
Prasinic acid (Elix et al. Reference Elix, Jones, Lajide, Coppins and James1984)=prasina C (sensu Coppins Reference Coppins1983): TLC 24; U2+, U3± white (“grey or pale mauve” or “colourless” in Coppins Reference Coppins1983, depending on solvent system used); H grey; U3 after charring dark blue-grey; extract soon milky by forming rod-shaped crystals.
-
Methylhiascic acid: TLC 12; U2+ grey, U3−; H grey; can only be separated from gyrophoric acid by TLC in solvent C (Orange et al. Reference Orange, James and White2010), not in A. It was found in Micarea borbonica sp. nov. and M. peliocarpa.
-
Unknown 1=unknown in M. melanoprasina sp. nov.: TLC 31; U2+, U3−; H pale greenish grey; U3 after charring light greenish grey; crystals melting on gentle heating, soon recrystallizing; extract clear, gummy, without crystals; a substance probably related to micareic acid.
-
Unknown 2=most probably confluentic acid, or a related compound, detected in M. takamakae sp. nov.: TLC 24; U2+, U3−; H pale yellowish with greyish rim; extract gummy, with radial wrinkles, later turning milky.
-
Unknown 3=unknown in M. pseudolignaria sp. nov.: TLC 28; U2+, U3−; H yellow-brown (as in methoxymicareic acid); U3 after charring dark brown (as in methoxymicareic acid); extract clear, gummy, white within the ring, amorphous, not polarizing; small crystals in thallus not melting.
Besides the well-known compound argopsin and a xanthone that we tentatively refer to as thiophanic acid, two compounds are worth mentioning: alectorialic acid, diagnostic for M. alectorialica sp. nov., and a fatty acid (protolichesterinic acid) reported here for the first time in the genus and diagnostic for M. sublithinella sp. nov. Another fatty acid, close to rangiformic acid, was also detected in M. alectorialica sp. nov.
Insoluble pigments have been characterized by Meyer & Printzen (Reference Meyer and Printzen2000); the following have been detected in our material (H being the reaction to HCl, and N to HNO3):
-
Cinereorufa-green: bright aeruginose, K+ green, H−, N+ red, found in Micarea alectorialica sp. nov., M. boryana sp. nov., M. incrassata, M. isidiosa sp. nov., M. lignaria, M. melanoprasina sp. nov., M. pseudocoppinsii sp. nov., M. pseudolignaria sp. nov., M. takamakae sp. nov. and M. tenuispora sp. nov. In M. borbonica sp. nov., M. cilaoensis sp. nov., M. cinerea, M. peliocarpa and M. pseudocoppinsii sp. nov., the same pigment is detected but has a slightly different colour (greyish blue and not bright aeruginose as in other species) and is a surface incrustation of hyphae and not diffuse in the hymenial gelatine (as in other species). We consider it a variant of the Cinereorufa-green that requires further investigation.
-
Laurocerasi-brown: pale reddish brown, K+ purple-brown, H−, N−, found in Micarea bebourensis sp. nov.
-
Sedifolia-grey: greyish, K+ violet, found in Micarea hedlundii and M. prasina.
-
Superba-brown: pale brown, K+ dull brown, found in Micarea incrassata.
-
Unknown 4: violet-red, K+ green, H−, forming dark blue granules in N which get more greenish when further applying K, found in the hymenium of Micarea isidiosa sp. nov. The granules are quite similar to those found in Lecidea hypnorum but, in this species, they do not require application of N to be detected.
-
Unknown 5: this substance can be detected at the goniocysts centre of Micarea hedlundii as tiny oily drops, which are not soluble in acetone, and therefore cannot be analyzed with TLC.
Additional brown pigment(s) that lack characteristic reactions and have thus not been classified by Meyer & Printzen (Reference Meyer and Printzen2000) were also observed.
In several species (Micarea bebourensis sp. nov., M. boryana sp. nov., M. levicula, M. melanoprasina sp. nov. and M. prasina), and in some collections of others (M. isidiosa sp. nov., M. lignaria and M. pseudolignaria sp. nov.), the thallus hyphae are I+ light blue after soaking in KOH (hemiamyloid); the reaction is often weak and not always constant. Such a hemiamyloid reaction has also been detected in many European species of Micarea but, to our knowledge, has never been found in other genera of the Lecanorales.
Taxonomy (Table 1)
Table 1. Summary of the taxonomy adopted for material of Micarea from Réunion and substantiation of the decision taken

We could detect and delimit 21 species in our material, out of which only eight can be assigned to an already described species; the other 13 are thus described as new to science in this paper. A key for their identification is presented below.
Two subgeneric entities yielded several new species and can be described as speciose groups on the island. They are the M. peliocarpa group and the M. prasina group (sensu Coppins Reference Coppins1983). The first one comprises M. borbonica sp. nov., M. boryana sp. nov., M. cinerea, M. peliocarpa and possibly M. pseudocoppinsii sp. nov., while the second includes M. levicula, M. melanoprasina sp. nov., M. prasina and possibly M. hyalinoxanthonica sp. nov. The latter group further includes material that contains methoxymicareic acid and belongs to M. micrococca s. lat. (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009); its taxonomy needs further study and is not dealt with in this paper.
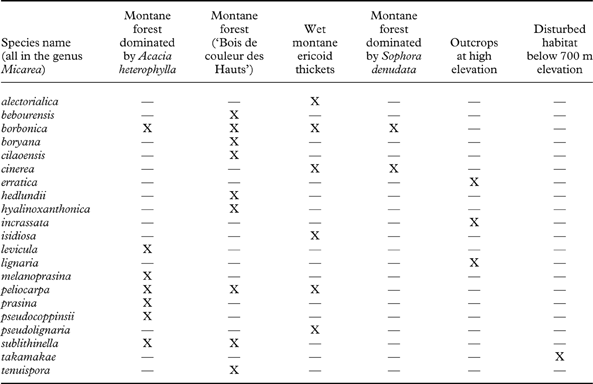
Ecology and biogeography (Table 2)
Table 2. Distribution of the representatives of the genus Micarea in the different habitats where they grow on Réunion
Interestingly, no species known so far only from the Southern Hemisphere was detected in our material, although one (M. boryana sp. nov.) seems to be close to Micarea mutabilis, described and as yet known only from Tasmania (Australia). The austral element detected in the working list of lichens present on Réunion (van den Boom et al. Reference van den Boom, Brand, Ertz, Kalb, Magain, Masson, Schiefelbein, Sipman and Sérusiaux2011) cannot thus be consolidated. The other species of Micarea so far restricted to the Southern Hemisphere (Coppins & Kantvilas Reference Coppins and Kantvilas1990; Kantvilas & Elix Reference Kantvilas and Elix1994; Coppins Reference Coppins1999; Aptroot Reference Aptroot2002; Fryday Reference Fryday2004; Cáceres et al. Reference Cáceres, Mota, de Jesus and Aptroot2013) are: Micarea almbornii Coppins, M. corallothallina M. Cáceres et al., M. endoviolascens Coppins, M. intersociella (Sirt.) Coppins, M. magellanica (Müll. Arg.) Fryday (=M. austroternaria Coppins & Kantvilas), M. pannarica Fryday, M. subgranulans (Vain.) Aptroot and M. subternaria (Vain.) Aptroot. Micarea poliocheila (Vain.) Aptroot, restricted to Brazil (Minas Gerais and Rio de Janeiro), has been transferred to Bilimbia by Kalb (Reference Kalb2007).
Interestingly, habitats at the highest elevations (>2000 m), and more precisely outcrops in dry and exposed conditions above cloud level, yielded three species that are either widespread in the temperate and boreal zones of the Northern Hemisphere (M. erratica and M. lignaria) or bipolar, being also found in subantarctic islands (M. incrassata). Such observations demonstrate the impressive dispersion capacity of lichen species (Honegger Reference Honegger1993; Romeike et al. Reference Romeike, Friedl, Helms and Ott2002).
Micarea is absent in all habitats at low elevations on the island, including the rocky shores, open and anthropogenic vegetation, recent or old lava flows, and remnants of low elevation rainforest dominated by the Sapotaceae (e.g. at the nature reserve of ‘Mare Longue’ near St-Philippe). Following the vegetation typology provided by Cadet (Reference Cadet1977), Strasberg et al. (Reference Strasberg, Rouget, Richardson, Baret, Dupont and Cowling2005) and Lacoste et al. (Reference Lacoste, Delbosc and Picot2011), we recognized six vegetation types that accommodate species of Micarea (Fig. 1):
-
Montane forest with large boles of Acacia heterophylla, either in natural stands or in plantations, at >1500 m elevation.
-
Montane forest known as ‘Bois de couleurs des Hauts’, easily characterized by the abundance of tree species in the genera Dombeya (Malvaceae), Monimia and Tambourissa (Monimiaceae), and others in the Euphorbiaceae, Rubiaceae, Rutaceae, Nuxia verticillata, and locally the native bamboo Nastus borbonicus and tree ferns (Cyathaea), mainly at elevations >1300 m.
-
Wet montane thickets with tall ericoid species including Erica reunionensis, with exuberant development of ground bryophyte cover (including species of Sphagnum), at c. 1700–1900 m.
-
Open and partially grazed forests, dominated by Sophora denudata, on the SW side of the Piton de la Fournaise, at c. 1800–2000 m.
-
Dry and exposed outcrops, at high elevations (>2000 m) with xerophytic bryophytes.

Fig. 1. Photographs of the main habitats for Micarea on Réunion. A, typical peeling trunk of Acacia heterophylla, near Savane Cimetière (NNW of Piton de la Fournaise): seven species of Micarea were found in this habitat; B, typical view of understorey of the montane ‘Bois de couleurs des Hauts’ (Forêt de Bébour): the richest habitat for Micarea with nine species; C, wet montane ericoid thickets with Erica reunionensis (along the track from Bélouve to Caverne Dufour): six species occur in this spectacular habitat, including one new species with soralia and producing alectorialic acid, and one new species with isidia; D, open forest dominated by Sophora denudata, along road to Bourg Murat (WNW of Piton de la Fournaise): only two species were found in this habitat, including the new M. borbonica, the most ubiquitous species on the island; E, exposed outcrops at high elevations near the Piton de la Fournaise: three species occur in such habitats: M. erratica, M. lignaria, both widespread in the Northern Hemisphere, and M. incrassata, a bipolar species; F, lava flow of April 2007 within the ‘Enclos’, photographed in May 2008: no species of Micarea were found on old or recent lava flows at low elevation. In colour online.
The numbers of species of Micarea found in each habitat are presented in Table 2. As expected, the most diverse habitat is the montane forest ‘Bois de couleurs des Hauts' with nine species, including five new species which have only been found here (M. bebourensis sp. nov., M. boryana sp. nov., M. cilaoensis sp. nov., M. hyalinoxanthonica sp. nov. and M. tenuispora sp. nov.). It is immediately followed by the Acacia heterophylla forests, with seven species. The soft, rather acidic, quick-drying and often peeling bark of that endemic tree seems to be a very suitable habitat for many species of Micarea. The wet ericoid thickets shelter six species, with two species new to science (M. alectorialica sp. nov. and M. isidiosa sp. nov.) being restricted to it. Furthermore, the Sophora open forest has only two species, including M. borbonica sp. nov. which is the only species present in all types of montane forest. The most distinctive M. takamakae sp. nov. is only known from a non-montane forest habitat.
Like many isolated islands and archipelagos all over the world, Réunion's original biota has been devastated by human impact and the local fauna and flora are much altered by exotic species; therefore conservation evaluation and planning require detailed studies of biodiversity processes (Lagabrielle et al. Reference Lagabrielle, Rouget, Payet, Wistebaar, Durieux, Baret, Lombard and Strasberg2009). In Réunion, the native vascular flora is estimated at c. 500 species and more than 3500 have been introduced, 62 being highly invasive (Baret et al. Reference Baret, Rouget, Richardson, Lavergne, Egoh, Dupont and Strasberg2006). The question may therefore arise regarding the status of lichen species observed on the island: are some species exotic? Although we have no clue at all to support this hypothesis, the question may be relevant for species growing on introduced tree species, such as Cryptomeria japonica, a species widely planted on Réunion. The case of three newly described species, M. boryana, M. cilaoensis and M. tenuispora, is illustrative as they have been detected only on trunks of Cryptomeria. However, the acidic and peeling bark of this tree makes it a suitable habitat for pioneer and fast-growing taxa such as Micarea, and is also easy to sample. We therefore postulate that these three species are native to Réunion. That Micarea grows easily on planted Cryptomeria is also observed in the Azores, an archipelago in the North Atlantic Ocean with a high rainfall level and a much degraded vegetation, where Cryptomeria is also widely planted (Purvis & James Reference Purvis and James1993).
Key to the species of Micarea in Réunion
-
1 Thallus with gyrophoric acid or alectorialic acid, C+ red ... 2
Thallus without gyrophoric and alectorialic acids, C− or C+ orange ... 12
-
2(1) Thallus with genuine convex soralia ... 3
Thallus not sorediate ... 4
-
3(2) Thallus with alectorialic acid (K+ yellow, P+ yellow) ... M. alectorialica
Thallus with gyrophoric acid (K−, P−) ... M. pseudocoppinsii
-
4(2) Thallus with, or entirely made of, isidiiform areolae, ascospores long and narrow (>20 µm long and <3 µm wide) ... 5
Thallus without isidiiform areolae ... 6
-
5(4) Ascospores bacillar, straight to slightly curved; aeruginose hymenium contrasting with reddish purple hypothecium ... M. isidiosa
Ascospores acicular; hymenium colourless and hypothecium pale brown to dark bluish ... M. tenuispora
-
6(4) Ascospores 0–1(–2)-septate ... 7
Ascospores 3-septate or more ... 9
-
7(6) Thallus formed of vivid green goniocysts; apothecia white ... M. levicula
Thallus not formed of goniocysts; apothecia white or grey to brown ... 8
-
8(7) Thallus thin, uneven; macroconidia 38–48 µm ... M. cilaoensis
Thallus tuberculate; macroconidia unknown ... M. bebourensis
-
9(6) Ascospores fusiform, mainly or only 7-septate ... 10
Ascospores mainly or only 3-septate ... 11
-
10(9) Ascospores 3·4–4·4 µm wide; macroconidia 57–78×1·2–1·5 µm ... M. cinerea
Ascospores 3·0–4·1 µm wide; macroconidia 30–40×1·0–1·3 µm ... M. borbonica
-
11(9) Ascospores fusiform, 3·6–4·7 µm wide, less than 17 µm long ... M. peliocarpa
Ascospores narrowly clavate to needle-like, 2·0–2·5 µm wide, more than 17 µm long ... M. boryana
-
12(1) Thallus with xanthones, C+ orange-yellow ... M. hyalinoxanthonica
Thallus without xanthones, C− ... 13
-
13(12) Thallus composed of goniocysts ... 14
Thallus not composed of goniocysts ... 18
-
14(13) Pycnidia stipitate, with tiny hairs, thallus with K+ violet pigment ... M. hedlundii
Pycnidia not stipitate, without hairs, or pycnidia absent ... 15
-
15(14) Thallus with conspicuous black prothallus; presence of Cinereorufa-green pigment in thallus and apothecia and unknown pigment in apothecia ... M. melanoprasina
Thallus without dark prothallus ... 16
-
16(15) Apothecia dark; micareic or confluentic acid present ... 17
Apothecia whitish to brownish; methoxymicareic acid present ... M. micrococca s. lat. (not treated)
-
17(16) Apothecia with K+ violet pigment; micareic acid present ... M. prasina
Apothecia without K+ violet pigment but epihymenium blue-green, K−; confluentic acid present ... M. takamakae
-
18(13) Ascospores (1–)3–7-septate ... M. lignaria
Ascospores 0–1-septate ... 19
-
19(18) Saxicolous; no lichen substances ... 20
Corticolous or terricolous; lichen substances present ... 21
-
20(19) Apothecia marginate, up to 0·4 mm diam.; epihymenium aeruginose ... M. erratica
Apothecia immarginate, up to 0·6 mm diam., epihymenium brown, locally with blue-green pigment ... M. incrassata
-
21(19) Ascospores 13–15×5–6 µm; protolichesterinic acid present ... M. sublithinella
Ascospores 8–9×3–4 µm; chemistry different, not a fatty acid ... M. pseudolignaria
The Species
Micarea alectorialica Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807670
Thallus areolate, sorediate, soralia to 0·6 mm diam.; apothecia convex, up to 0·7 mm, shining black; ascospores fusiform, 17–20×4·5–5·6 µm, (1–)3-septate; production of alectorialic acid.
Type: Réunion, Forêt de Bébour, path from Bélouve to cabane Dufour, 21°5·10′S, 55°31·36′E, 1890 m, wet montane ericoid thickets, on Erica, 2 June 2008, M. Brand 58863 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2A)

Fig. 2. Photographs of the new species described from Réunion. A, Micarea alectorialica sp. nov. (holotype); B, M. bebourensis sp. nov. (holotype); C, M. borbonica sp. nov. (holotype); D, M. boryana sp. nov. (holotype); E, M. cilaoensis sp. nov. (holotype); F, M. hyalinoxanthonica sp. nov. (holotype); G, M. isidiosa sp. nov. (holotype); H, M. pseudocoppinsii sp. nov. (holotype). Scale=1 mm. In colour online.
Thallus c. 5 cm across, formed by dispersed areolae; areolae first globose, 0·1–0·2 mm across, pale-coloured, then bursting into a soralium; soralia to 0·6(–0·8) mm diam., convex, sometimes aggregated to almost stipitate, white to light greenish; soredia 25–35 µm. Photobiont micareoid, cells c. 5·0–5·5 µm. Cortex absent, but in young areolae upper layer with more crystals; soredia strongly inspersed with crystals (alectorialic acid).
Apothecia up to 0·7 mm diam., shining black, convex without margin, often complex, and then made of fused small apothecia. Excipulum bluish in outer parts, inside pale, formed by strongly conglutinated radiating hyphae. Hypothecium pale or light bluish. Hymenium c. 55 µm high; epihymenium dark blue. Paraphyses c. 1·5 µm thick, apically branched and anastomosing. Asci clavate, c. 46×12 µm, tholus of the Micarea-type. Ascospores 8 per ascus, fusiform 17·0–20·0× 4·5–5·6 µm, (1–)3-septate.
Micropycnidia numerous, superficial between areolae, black, c. 70 µm; microconidia narrow, fusiform with tapering ends, 6·6–8·3×0·9–1·0 µm.
Chemistry
Alectorialic acid (K+ yellow, P+ yellow, C+ red) in thallus and soralia; fatty acid also present (extract as rangiformic acid, but larger agglomerations). Pigment in thallus and apothecia belonging to Cinereorufa-green.
Etymology
The new species is named after the chemical compound that makes this species easy to recognize amongst the species of Micarea present on Réunion.
Habitat and distribution
On Erica stems at 1900–2000 m, in wet montane ericoid thickets, abundant.
Notes
Alectorialic acid is rare amongst species of Micarea as it is so far detected in only two species: M. magellanica (Müll. Arg.) Fryday (=M. austroternaria Coppins & Kantvilas) restricted to the Southern Hemisphere (Coppins & Kantvilas Reference Coppins and Kantvilas1990; Fryday Reference Fryday2004), and M. submilliaria (Nyl.) Coppins, a mostly Northern Hemisphere species (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009), but both do not produce soralia, although the surface of the latter is usually eroding to form sorediate patches. Only three other species of Micarea produce soralia: M. coppinsii Tønsberg, M. pseudocoppinsii (here described as new) and M. viridileprosa Coppins & van den Boom (soralia usually not clearly delimited and forming a leproid surface); they can all be distinguished by the production of gyrophoric acid (van den Boom & Coppins Reference van den Boom and Coppins2001; Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009).
Additional specimen examined. Réunion: same locality as the type, 21°05′31″S, 55°30′41″E, 1910–2000 m, wet ericoid thickets with E. reunionensis and Phylica nitida, on Erica, 9 xi 2009, N. Magain & E. Sérusiaux s. n. (LG).
Micarea bebourensis Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807671
Thallus typically tuberculate when young, tubercles 0·10–0·15 mm across; apothecia up to 0.6 mm, greyish brown; ascospores ovate to shortly bacillar, 11–14× 3·3–4·3 µm, 1(–3)-septate; production of gyrophoric acid.
Type: Réunion, Forêt de Bébour, trail to Cassé de Takamaka, 21°07·5′S, 55°34·5′E, 1340 m, wet montane rainforest, on rotting standing trunk, 1 June 2008, P. van den Boom 40344 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2B)
Thallus up to 2 cm diam., pale to brownish greyish or greenish, uneven, typically tuberculate when young, with tubercles 0·10–0·15 mm broad and high, eventually formed of coarse and agglomerated granules, first pale, eventually breaking up at the top of tubercules or at the margins of granules, and forming a rough blue-grey surface (resembling tiny soralia, but no genuine soredia are formed). Cortex absent, often with thin, clear epinecral layer. Photobiont micareoid. Thallus filled with crystals (gyrophoric acid).
Apothecia up to 0·6 mm diam, light to dark greyish brown, often unevenly coloured (partly dark, partly pale), almost globose, with constricted base. Excipulum greyish brown in outer parts, pale inside. Hypothecium pale (reddish) brown, conglutinate. Hymenium c. 50–60 µm high; epihymenium patchily brownish. Paraphyses 0·7–1·1 µm thick, branched and strongly anastomosing; some paraphyses with a thickened apex (1·8 µm). Asci cylindrical, c. 40–53×10 µm, tholus of the Psora-type. Ascospores 8 per ascus, ovate to shortly bacillar, 11–14×3·3–4·3 µm, 1(–3)-septate.
Micropycnidia inconspicuous, immersed, with brownish top, c. 35 µm. Microconidia bacillar with rounded ends, some slightly curved, 5·9–7·1×0·9–1·1 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red), mainly in the thallus, epihymenium, excipulum and pycnidia wall. Brownish pigment belonging to Laurocerasi-brown, in epihymenium, slightly brownish, K+ purple-brown; subhymenium and hymenium in K slightly rose-coloured.
Etymology
This new species is named after the Bébour forest, one of the largest and best preserved montane forests on Réunion, where many interesting lichen species grow.
Habitat and distribution
On introduced tree species, including Cryptomeria japonica and decaying wood, in lower montane forest at 1200–1500 m.
Notes
The almost globose apothecia, sometimes almost stipitate, and the thallus tubercles (best observed in young thalli) represent the most obvious characters to distinguish this species. Its generic assignment to Micarea is questionable as the asci are cylindrical (not clavate as in other species of the core group of Micarea) and the tholus is much akin to the Psora-type (sensu Ekman et al. Reference Ekman, Andersen and Wedin2008). The anatomy of apothecia (globose apothecia with a constricted base and development of excipular tissue) makes it close to Micarea lignaria and M. pseudolignaria sp. nov.; M. bebourensis is easily differentiated from both by the absence of an aeruginose pigment and the production of gyrophoric acid.
Additional specimens examined. Réunion: same locality as the type, P. van den Boom 40370 (h); Bélouve, 21°3·74′S, 55°32·29′E, 1500 m, montane forest, on planted Cryptomeria, 2008, M. Brand 58215 (h); Plaine-des-Palmistes, Ravine Sèche, 21°8·63′S, 55°35·31′E, 1200 m, planted exotic trees in sheltered valley, on Cryptomeria, 2008, M. Brand 58141 (h); Forêt de Bébour, path W of Col de Bébour, 21°07·2′S, 55°33·6′E, 1490 m, Cryptomeria plantation, on Cryptomeria, 2008, P. van den Boom 40322 (h).
Micarea borbonica Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807672
Thallus small, to 1·5 cm, dull white to pale greenish; apothecia up to 0·5 mm diam., whitish to pale brownish or pale to dark bluish grey; ascospores fusiform with rounded ends, 15–27×3·0–4·1 µm, 3–7-septate; macroconidia strongly and repeatedly curved sigmoid, 30–42×1·0–1·3 µm, indistinctly 3-septate; production of gyrophoric acid.
Type: Réunion, WNW of Piton de la Fournaise, along road to Bourg Murat, 21°11·9′S, 55°36·9′E, 1970 m, small forest with Sophora denudata trees, on Sophora, 4 June 2008, P. van den Boom 40644 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2C)
Thallus small, to 1·5 cm across, dull white to pale greenish, uneven, c. 50 µm high or consisting of small granules, some of them resembling small isidia. Upper part c. 15 µm without algae, of loosely interwoven hyphae. Photobiont micareoid, cells c. 5 µm, photobiont layer inspersed with crystals (gyrophoric acid).
Apothecia up to 0·2–0·5 mm diam., whitish to pale brownish or pale to dark bluish grey, often coloured only partially, with pale margin, flat or slightly convex to almost globose, adnate with constricted base. Excipulum pale or bluish grey in outer parts, pale inside. Hypothecium colourless. Hymenium c. 55 µm high; epihymenium colourless or partly blue-grey. Paraphyses 1·3–1·6 µm thick, branched and anastomosing. Asci clavate, 33–40×12–15 µm, tholus of the Micarea-type. Ascospores 8 per ascus, fusiform with rounded ends, 15–27×3·0–4·1 µm, (3–)7-septate, often curved.
Macropycnidia not rare, globose, 100–140 µm, top often blue-grey; macroconidia strongly and repeatedly curved sigmoid (when observed under unpressed cover slip), 30–42×1·0–1·3 µm (measured under pressed cover slip), faintly 3-septate. Micropycnidia inconspicuous, semi-superficial, c. 35 µm, pale. Microconidia narrow fusiform with tapering ends, 5–7(–10)×0·8–1·0 µm.
Chemistry
Gyrophoric and methylhiascic acids (K−, P−, C+ red), mainly in thallus, hymenium, excipulum and pycnidia. Pigment in apothecia belonging to Cinereorufa-green.
Etymology
The name of this new species refers to the old name of the island, ‘Ile Bourbon’ in French. It is indeed characteristic of all habitats, natural or anthropogenic, in the montane forest zone of the island.
Habitat and distribution
On native and exotic trees (Acacia heterophylla, Cryptomeria, Sophora denudata, Platanus and on branches of Erica), and decorticated wood, between 800–2000 m.
Notes
Ascospores in this new species are typically 7-septate, although 3–6-septate ones can be found usually in immature or poorly developed apothecia. Micarea borbonica is closely related to M. peliocarpa, a species also found in Réunion; it differs in having convex to subglobose areoles, longer and narrower ascospores (13·6–17·0×3·6–4·7 µm in collections of M. peliocarpa from Réunion), and is usually 7-septate (mostly 3-septate in M. peliocarpa). A further related species is M. cinerea, also found on Réunion, that can be distinguished by larger spores (24–30×3·4–5·0 µm) and much longer macroconidia (57–78×1·2–1·5 µm). See further notes under M. cinerea and M. peliocarpa.
Additional specimens examined. Réunion: Piton de la Fournaise, 0·5 km NW of Gîte du Volcan, 21°12·07′S, 55°41·53′E, 2040 m, mixed wood on N-slope, on Acacia heterophylla, 2008, M. Brand 58361, 58376 (h); Plaine-des-Palmistes, 21°6·98′S, 55°37·13′E, 1100 m, Platanus branches in garden, 2008, M. Brand 58696 (h); ibid., on decorticated wood of telegraph pole, 2008, P. van den Boom 40621 (h); Cilaos, Forêt du Grand Matarum, path to Cabane Dufour, 21°7·08′S, 55°29·19′E, 1770 m, wet montane ericoid thickets on steep SW slope, on thin twigs, 2008, M. Brand 59713 (h); WNW of Piton de la Fournaise, along road to Bourg Murat, 21°11·6′S, 55°37·5′E, 2090 m, disturbed wood with Sophora denudata, on Sophora, P. van den Boom 40665 (h); ESE of Le Tampon, NNE of Petite-île, 21°18·9′S, 55°35·4′E, 800 m, open place with mature Cryptomeria trees, on Cryptomeria, 2008, P. van den Boom 40806 (h).
Micarea boryana Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807673
Thallus areolate, greenish to greyish brown, eroding when old; apothecia to 0·4(–0·6) mm, grey to bluish black; ascospores narrowly clavate to needle-like, 16–27×2·0–2·5 µm, (1–)3(–4)-septate; production of gyrophoric acid.
Type: Réunion, Cirque de Cilaos, N of Cilaos, Forêt du Grand Matarum, trail to Caverne Dufour, 21°07·3′S, 55°29·2′E, montane forest, 1420 m, on Cryptomeria, 31 May 2008, P. van den Boom 40264 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2D)
Thallus up to 5 cm across, greenish to greyish brown, continuous, made of small flat to convex or even subglobose areolae, with a thin endo- to epiphloeodal dark bluish black prothallus made of narrow hyphae, colourless or partly dark brownish or bluish hyphae. Areolae either flat (0·1–0·6 mm broad, 0·05 mm thick) or subglobose (granules c. 0·1 mm across), eroding at the margins in old thalli, slightly shiny. Cortex absent, but a thin, clear epinecral layer can be observed. Photobiont micareoid, cells c. 5–8 µm. Thallus filled with crystals of gyrophoric acid.
Apothecia up to 0·4(–0·6) mm diam., grey to bluish black, sometimes with a pale margin, first flat, without a distinct margin, then semiglobose. Excipulum with radiating hyphae, varying in colour (often in same apothecium) from pale to superficially bluish and centrally light brown. Hypothecium pale or brownish. Hymenium c. 30–55 µm high; epihymenium patchily greyish-bluish. Paraphyses 1·0–1·2 µm thick, branched and anastomosing. Asci 25–37×10–15 µm, tholus of the Micarea-type. Ascospores 8 per ascus, in one bundle, narrowly clavate to needle-like, 16·0–27·0× 2·0–2·5 µm, (1–)3(–4)-septate, sometimes apically slightly enlarged and tapering towards their basal ends.
Mesopycnidia often present and conspicuous, sessile, c. 160–230 µm diam. and 200–270 µm in height, dark brownish, with white blob of conidia on top; pigment brown to greenish brown or aeruginose, K−; wall with crystals of gyrophoric acid, mainly near the apex. Mesoconidia ovate, 4·0–4·5×(1·3–)1·5–1·6 µm. Micropycnidia rarely present, inconspicuous, with a dark brownish wall. Microconidia fusiform, 4·0–4·5(–5·0)×1·0–1·1 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red) detected in thallus, but also in pale parts of apothecia and in mesopycnidia. Pigment in thallus and apothecia belonging to Cinereorufa-green. Brown pigment in hypothecium K+ reddish to purple-brown.
Etymology
This new species is named after the French officer and first lichen collector on the island, J. B. G. M. Bory de Saint-Vincent (1778–1846) who described several spectacular and now well-known species, such as Cladonia candelabrum, C. giganteum, Lobaria retigera, Stereocaulon vulcani and Sticta ambavillaria.
Habitat and distribution
Found at several localities on the island, between 1200–1500 m, always epiphytic on the exotic Cryptomeria japonica.
Notes
We first assigned these collections to Micarea mutabilis Coppins & Kantvilas, described from Tasmania (Coppins & Kantvilas Reference Coppins and Kantvilas1990). According to the original description, the latter, however, clearly differs by several important characters: absence of prothallus, larger asci (40–50×9–10 µm), ascospores longer and more septate [21–41× 2·2–3·0 µm; (1–)5–7(–8)-septate], absence of conspicuous pycnidia producing mesoconidia (the mesoconidia described for that species are actually microconidia). We believe such discrepancies are worth species recognition. Other species with needle-shaped or acicular ascospores are (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009): Micarea globulosella (Nyl.) Coppins, distinguished by its black and subglobose apothecia and smaller ascospores [(10–)12–19(–24)× 2·0–2·5(–3·0) µm]; M. synotheoides (Nyl.) Coppins distinguished by the absence of chemical compounds and variable ascospores [needle-like, fusiform to rod-shaped, (0–)1–3-septate, 14–35(–43)×1·8–2·5(–3·0) µm]; M. pycnidiophora Coppins & P. James, together with M. stipitata Coppins & P. James, both being easily recognized by their numerous, stipitate pycnidia; and finally M. tenuispora sp. nov., described here as new from Réunion, which is easily recognized by its isidiiform thallus areolae.
Additional specimens examined. Réunion: same locality as the type, 2008, P. van den Boom 41041 (h); Plaine-des-Palmistes, Ravine Sèche, 21°8·63′S, 55°35·31′E, 1200 m, planted exotic trees in wood in sheltered valley, on Cryptomeria, 2008, M. Brand 58141, 58146 (h), P. van den Boom 39776 (h); Bélouve, 21°3·74′S, 55°32·29′E, 1500 m, disturbed montane forest, on Cryptomeria, 2008, M. Brand 58206 (h).
Micarea cilaoensis Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807675
Thallus thin, uneven, consisting of slightly convex granules, green; apothecia to 0·4 mm, adnate, with constricted base; ascospores ovoid to ellipsoid, 11–12× 4·0–4·4 µm, 0–1-septate; production of gyrophoric acid.
Type: Réunion, Cirque de Cilaos, Forêt du Grand Matarum, trail to Caverne Dufour, 21°07·3′S, 55°29·2′E, 1420 m, montane forest, on Cryptomeria, 31 May 2008, P. van den Boom 40230 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2E)
Thallus rather extensive (3 cm across), green, thin (65–90 µm), uneven, made of slightly convex granules. Cortex absent. Photobiont micareoid, cells c. 5–9 µm. Photobiont layer inspersed with crystals of gyrophoric acid.
Apothecia up to 0·4 mm diam., whitish to greyish with pale margin, flat or slightly convex, adnate with constricted base. Excipulum well developed, made of thin reticulate hyphae, filled with crystals, colourless. Hypothecium colourless. Hymenium c. 35–42 µm high; epihymenium colourless or faintly greenish grey, in K unchanged or locally brownish. Paraphyses 1·3 µm thick, branched and anastomosing. Asci clavate, c. 28×12 µm, tholus of the Micarea-type. Ascospores 8 per ascus, ovoid to ellipsoid, 11–12×4·0–4·4 µm, 0–1-septate.
Pycnidia not rare, superficial or semi-immersed, white, ampulliform, c. 150 µm diam., top with crystals; macroconidia straight or more or less curved, 38–48×0·9–1·1 µm, non-septate, cylindrical with rounded ends; in same pycnidia, microconidia can be found: microconidia bacilliform, 5·2–7·1× 0·9–1·1 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red) detected in thallus, hymenium, excipulum, pycnidia. Pigment in apothecia belonging to Cinereorufa-green.
Etymology
The name chosen for this new species refers to the most spectacular cirque on the island, the Cirque de Cilaos, where this rare species has been discovered.
Habitat and distribution
Known from a single locality, on the exotic Cryptomeria japonica at the margin of disturbed natural forest, at 1420 m elevation.
Notes
Micarea cilaoensis could be confused with a species of Lecania because of its apothecia with a brownish disc and whitish margin, and its well-developed excipulum. However, examination of the hamathecium and ascus-type immediately point to the genus Micarea. Also remarkable are the large ampulliform pycnidia producing macro- and microconidia together. Micarea cilaoensis is close to M. denigrata (Fr.) Hedl., a widespread species in the Northern Hemisphere which also produces large ampulliform pycnidia; the latter differs by its narrower ascospores [(7–)9–16(–18)×2·0–3·3(–3·5) µm] and much smaller macroconidia (12–24×c. 1 µm; Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009).
Additional specimen examined. Réunion: same locality as the type, P. van den Boom 40229 (h, hb Brand 62255).
Micarea cinerea (Schaer.) Hedl.
Thallus up to c. 1 cm across, made of small granules (c. 0·1–0·2 mm diam.), some of them resembling small isidia, white to pale grey.
Apothecia to 0·3 mm diam., whitish, beige, pale yellowish brown or dark bluish grey, often unevenly coloured, with a slightly pale margin, flat or convex to almost globose, adnate with constricted base. Hymenium c. 50 µm; epithecium yellowish or more rarely bluish, K−. Hypothecium hyaline. Asci 37–45×15–20 µm, tholus of the Micarea-type. Ascospores 8 per ascus, fusiform, 24–30×3·4–5·0 µm, (3–)7-septate, straight to often curved.
Pycnidia large, c. 200 µm diam., partly immersed in areoles. Macroconidia long and slightly flexuose, 7–10-septate, 57–78×1·2–1·5 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red), mainly in thallus, hymenium, excipulum and pycnidia wall; methylhiascic acid absent. Pigment in apothecia belonging to Cinereorufa-green.
Habitat and distribution
On Erica in wet montane ericoid thickets at 2000 m, and on trunk of Sophora at similar elevation.
Notes
In European collections of this species, methylhiascic acid is detected as well as gyrophoric acid; this acid has not been detected in the material from Réunion. See further data under M. borbonica. Micarea cinerea is a widespread species as it occurs in Europe, North and Central America, Asia and Australia (Tasmania), according to Coppins (Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009).
Specimens examined. Réunion: Forêt de Bébour, trail from Gîte de Bélouve, 3·5 km to the south-west, to Caverne Mussard, 21°05·3′S, 55°31·3′E, 1980 m, wet montane ericoid thickets, 1980 m, on Erica, 2008, P. van den Boom 40526, 40555 (h, hb Brand 61471); WNW of Piton de la Fournaise, along road to Bourg Murat, 21°11·9′S, 55°36·9′E, small open forest with Sophora denudata, on Sophora, 1970 m, 2008, P. van den Boom 40644A (h).
Micarea erratica (Körb.) Hertel et al.
Thallus of scattered whitish areoles; areolae up to 0·5 mm diam., with a c. 15 µm thick epinecral layer. Photobiont not micareoid, cells to 15 µm.
Apothecia up to 0·4 mm broad, black, flat, with a distinct margin. Excipulum of conglutinated radiate hyphae, outer part aeruginose, inner part hyaline. Hypothecium dark brown. Hymenium c. 25 µm; epihymenium aeruginose. Paraphyses simple, c. 1·8 µm thick. Asci c. 25×10 µm, tholus of the Micarea-type. Ascospores 8 per ascus, narrowly ellipsoid, c. 7×3 µm.
Mesopycnidia numerous, c. 70–100 µm, immersed, upper side dark bluish, lower side unpigmented. Mesoconidia c. 3·2–4·5× 1·1–1·9 µm.
Chemistry
No lichen substances detected.
Habitat and distribution
On volcanic rocks in meadow, at c. 2000 m elevation.
Notes
This single collection falls within the variation of European specimens of that species, including the size and height of apothecia, and we have no doubt it belongs to the widespread M. erratica. The species has been recognized as the sole representative of the new genus Leimonis (Harris Reference Harris2009), an option not retained so far (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009).
Specimen examined. Réunion: track Bourg Murat to Piton de la Fournaise, 21°12·63′S, 55°36·57′E, 1970 m, volcanic rock outcrops in meadow on NW slope, on stones, 2008, M. Brand 59027 (h).
Micarea hedlundii Coppins
Thallus less than 1 cm across, made of abundant and aggregated goniocysts, forming coralloid masses, dark greenish to greyish. Goniocysts 17–32 µm diam., locally with coarse crystals. Hyphae partly with brown-grey pigment (K+ violet). Photobiont micareoid, c. 5 µm.
Apothecia absent.
Pycnidia stipitate; stipe 0·24–0·40 mm high, 100–160 µm thick, surface with loose, curly, grey tomentum, made of c. 1·7 µm thick hyphae; pycnidia c. 140 µm, 1–3(–5) on a stipe. Mesoconidia elliptic, c. 4·2–5·2× 1·6–2·1 µm.
Chemistry
Unknown 5 at the centre of goniocysts, and Sedifolia-grey.
Habitat and distribution
On soft rotting wood of standing trunks in montane forest, at 1400–1900 m elevation.
Notes
Apothecia are absent in our material from Réunion, but otherwise we could not detect any differences with available collections from Western Europe, from the Albertine Rift in Africa and most recent descriptions (Czarnota Reference Czarnota2007; Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009). The recently described M. tomentosa Czarnota & Coppins (Czarnota Reference Czarnota2007), known from central and northern Europe (Suija et al. Reference Suija, Lõhmus and Motiejūnaitė2008), also has stalked pycnidia but differs by the complete absence of pigment H in the goniocysts.
Specimens examined. Réunion: Cilaos, Forêt du Grand Matarum, along path to Cabane Dufour, 21°6·29′S, 55°29·58′E, 1870 m, low forest on steep SW slope, on Dombeya tree, 2008, M. Brand 59739 (h); Forêt de Bébour, trail to Takamaka, 21°05·9′S, 55°34·3′E, 1370 m, wet rainforest, on rotting standing trunk, 2008, P. van den Boom 40883 (h).—Rwanda: Virunga volcanoes, southern flank of the Karisimbi, c. 3400 m, open forest with Hagenia abyssinica and Hypericum revolutum, on big bole of Hagenia, 1974, J. Lambinon 74/1524 (LG; confirmed by B. J. Coppins, 1992); ibid., 01°29′12·56″S, 029°28′37·38″E, c. 3200 m, on Hagenia, x 2010, E. Sérusiaux s. n. (LG).
Micarea hyalinoxanthonica Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807676
Thallus thin, pale greenish, made of goniocysts; apothecia up to 0·6 mm, adnate pale beige, translucent when wet; ascospores bacillar, 13·2–17·0×3·2–3·6 µm, 3-septate; production of xanthone (thallus and apothecia C+ orange).
Type: Réunion, Forêt de Bébour, Sentier de Takamaka, 21°06·48′S, 55°34·02′E, 1380 m, low trees in montane forest, on Dombeya, 7 June 2008, M. Brand 59293 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2F)
Thallus c. 3 cm diam., pale greenish, thin, overgrowing decaying bryophytes, soft, felty, made of small (20–30 µm diam.) goniocysts. Photobiont micareoid, 5–8 µm, individually surrounded with a mantle of furcate hyphae, partly aggregated in goniocysts; thallus also with strait, thick-walled hyphae (c. 2 µm) and sometimes including crystals.
Apothecia abundant, 0·1–0·5(–0·6) mm diam., adnate with constricted base, pale beige, translucent when wet, rarely greyish, first flat, with hardly distinct margin, then convex. Excipulum thin, chondroid, with thin, branching hyphae, outer parts sometimes with protruding hyphae, giving a hairy appearance. Hypothecium pale or patchily light brownish. Hymenium c. 50 µm high, strongly conglutinated, colourless. Paraphyses c. 1·3 µm thick, branched and anastomosing. Asci clavate, c. 45–50×12 µm; tholus of Micarea-type. Ascospores 8 per ascus, bacillar, 13·2–17·0×3·2–3·6 µm, 3-septate, distinctly constricted at septa, with more or less globose cells.
Pycnidia absent.
Chemistry
Xanthone (thiophanic acid?) in thallus and apothecia (K+ yellow; P−, C+ and KC+ orange). Crystals in thallus and apothecia, in streaks in hymenium and excipulum.
Etymology
The name chosen combines the chemical compound produced by this new species and a diagnostic character of its apothecia, being translucent when wet.
Habitat and distribution
Only known from the type locality. It grows on decaying liverworts on bole of a Dombeya in rainforest (c. 1400 m).
Notes
Young globose apothecia of no more than 100 µm diam. contain already ripe asci. TLC gives the same result as with Micarea xanthonica Coppins & Tønsberg, which contains thiophanic acid (Coppins & Tønsberg Reference Coppins and Tønsberg2001). This species can be distinguished by its thallus entirely made of dense goniocysts, very similar to that of M. prasina, apothecia rare or even absent, and ascospores (0–)1(–3)-septate. A further species containing xanthone is Micarea isabellina Coppins & Kantvilas, so far only known from Tasmania, growing on rocks or terricolous (Coppins & Kantvilas Reference Coppins and Kantvilas1990). It can be distinguished by its areolate to irregularly warty thallus, black apothecia and larger ascospores (19–26× 3·5–4·0 µm). Furthermore, M. hyalinoxanthonica can be distinguished by its ascospores that are distinctly constricted at septae, with more or less globose cells.
Micarea incrassata Hedl.
Thallus c. 2 cm across, composed of dispersed to adjacent areolae on rock, overgrowing mats of cyanophyta (Stigonema); areolae beige greenish, globose to convex, 0·1–0·2 mm. Photobiont micareoid, cells c. 5 µm. Cortex absent, thallus without crystals. Genuine cephalodia absent, but thallus clearly associated with Stigonema, the cells of which are clearly intermingled with hyphae within the areolae.
Apothecia up to 0·6 mm diam., black, convex without margin. Excipulum hardly distinct from hymenium. Hypothecium dark brown. Hymenium c. 40 µm high; epihymenium brown, locally with blue-green pigment. Paraphyses c. 1·8 µm thick, mostly simple, occasionally branched. Asci clavate, c. 27–30×8–10 µm, tholus of the Micarea-type. Ascospores 8 per ascus, ellipsoid, 8·6–10·0×4·2–5·0 µm, non-septate, wall c. 0·3 µm thick.
Micropycnidia scarce, between areolae, wall dark brown, c. 60 µm; microconidia bacillar, 5·7–6·8×1·0–1·1 µm.
Chemistry
No lichen substance. Pigment in thallus and apothecia belonging to Cinereorufa-green, pigment in hypothecium belonging to Superba-brown.
Habitat and distribution
Only known from a single collection, on recently exposed volcanic rock at high elevation.
Notes
Our single collection is very close to the European material of Micarea incrassata that we could examine. It differs, however, by its constantly 0-septate ascospores (vs. 0–2-septate in European material), smaller asci and thus lower hymenium, and absence of a chemical compound (European material has thallus crystals, not dissolving in K or HCl, but slowly in acetone). Pending further studies, we don't consider these variations to be worth taxonomic recognition.
Micarea incrassata is a further example of a species widespread in the Northern Hemisphere and also detected in Australia (New South Wales) and subantarctic islands, and that occurs patchily on summits in tropical areas. A similar example is Fuscopannaria praetermissa, present on high mountains in East Africa and in Réunion (van den Boom et al. Reference van den Boom, Brand, Ertz, Kalb, Magain, Masson, Schiefelbein, Sipman and Sérusiaux2011).
Specimen examined. Réunion: Piton de la Fournaise, S slope of Pas de Bellecombe, 21°12·6′S, 55°41·8′E, 2250 m, recent volcanic rock on steep S-slope, 2008, M. Brand 58333 (h).
Micarea isidiosa Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807677
Thallus formed of isidioid granules growing on partly dark prothallus, greenish to brownish; apothecia up to 0·4 mm in diam, black; hypothecium purplish or reddish; ascospores bacillar, straight to slightly curved, 22·5–29·8×2·8–2·9 µm, 3–7-septate; production of gyrophoric acid.
Type: Réunion, Forêt de Bébour, path from Bélouve to cabane Dufour, 21°05·10′S, 55°31·36′E, 1890 m, wet montane ericoid thickets, on Erica, 2 June 2008, M. Brand 58862 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 2G)
Thallus up to 3 cm diam., greenish to brownish, discontinuous, made of small globose or isidia-like areolae on thin endo- to epiphloeodal prothallus with narrow colourless or partly brownish or bluish hyphae. Areolae first granular (0·1 mm across) eventually becoming isidiiform (0·30×0·05 mm), unbranched or forked. Cortex absent. Photobiont micareoid. Thallus filled with crystals (gyrophoric acid).
Apothecia up to 0·4 mm diam., black, first flat, without distinct margin, then semiglobose. Excipulum with radiating hyphae, purplish, with few crystals. Hypothecium reddish or bluish. Hymenium c. 40 µm high; epihymenium and hymenium intense blue-green. Paraphyses c. 1·4 µm thick, branched. Asci c. 30–34×10 µm, tholus of the Micarea-type. Ascospores 8 per ascus, bacillar, straight to slightly curved, 22·5–29·8×2·8–2·9 µm, 3–7-septate.
Micropycnidia inconspicuous, immersed, with dark bluish wall. Microconidia fusiform, 3·7–4·1×0·9–1·0 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red), mainly in thallus, but also detected in apothecia. Blue pigment in thallus and apothecia K+ intense green, N+ red and H+ darker, most probably belonging to Cinereorufa-green; red pigment mainly present in hymenium (unknown 4) K+ green, N producing a precipitate of dark blue granules, H−.
Etymology
The name refers to the isidioid areolae forming the thallus.
Habitat and distribution
On Erica stems in wet ericoid thickets at elevations between 1900–2000 m.
Notes
The thallus of this species seems to be isidiate; however, the ‘isidia’ are not outgrowths on a thallus or areolae as they grow directly on the prothallus; they thus represent isidiiform areolae. The thallus and apothecia of this species are much akin to those of M. tenuispora, also described as new in this paper; the bacillar ascospores, more vivid colours in apothecium section, with an aeruginose tinge in the epihymenium contrasting with a reddish purple hypothecium, and the unique precipitate of dark blue granules in N in the hymenium are the diagnostic characters.
Additional specimens examined. Réunion: Forêt de Bébour, path from Bélouve to cabane Dufour, 3 km from Gîte Bélouve, 21°05·10′S, 55°31·36′E, 1890 m, tall ericoid thickets in montane forest, on Erica, 2008, M. Brand 58865 (h); ibid., 21°05·3′S, 55°31·3′E, 1980 m, on Erica, 2008, P. van den Boom 40529 (h).
Micarea levicula (Nyl.) Coppins
Thallus diffuse, vivid green, made of delicately and finely coralloid goniocysts; goniocysts 25–53 µm diam. or cylindrical (c. 50× 18 µm), round to ovate, often more or less angular, fused to form branched coralloid structures up to 150 µm high, made of conglutinated hyphae and micareoid photobiont (cells c. 6 µm), inspersed with crystals (gyrophoric acid), locally some hyphae protrude to form low papillae on the surface.
Apothecia up to 0·5 mm diam., white, adnate, slightly constricted at base, without any visible margin, convex. Excipulum narrow (<10 µm thick), formed by radiating, conglutinate hyphae; surface of excipulum sometimes rough because of protruding hyphae. Hypothecium colourless. Hymenium 50–55 µm high. Paraphyses 1·3–1·6 µm thick, branched and anastomosing. Asci c. 39–55×8–12 µm, tholus of the Micarea-type. Ascospores 8 per ascus, ellipsoid, 10·3–10·8×3·7–4·1 µm, 0–1-septate.
Micropycnidia occasionally present, small, white; microconidia 9·8×0·8 µm, narrow fusiform. Mesoconidia occasionally present, 5·5×1·3 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red), crystals present only at the centre of the goniocysts.
Habitat and distribution
On natural stands of Acacia heterophylla; so far only found near the Piton de la Fournaise, where it grows with the related M. prasina.
Notes
Micarea levicula has been recognized as a member of the M. prasina group by van den Boom & Coppins (Reference van den Boom and Coppins2001), on the basis of its type and only available collection from Cuba (H-Nyl. 20762). The short description provided matches the material collected in Réunion. Micarea viridileprosa Coppins & van den Boom, also a member of the prasina group with gyrophoric acid, differs by the loose, non-coralloid and smaller, more soredia-like goniocysts, the localization of the gyrophoric acid crystals on the outer parts of the hyphae surrounding the goniocysts, and finally by the narrower spores (c. 11–14× 3·0–3·4 µm). Micarea viridileprosa is so far known from western Europe, where it can be quite common, and Tasmania (van den Boom & Coppins Reference van den Boom and Coppins2001; Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009). The recently described M. corallothallina (Cáceres et al. Reference Cáceres, Mota, de Jesus and Aptroot2013) from NE Brazil has a thallus made of “irregularly densely branched isidioid granules of c. 50 µm thick” but can be distinguished by the absence of any chemical coumpounds.
Specimens examined. Réunion: NNW of Piton de la Fournaise, trail along Ravine Savane Cimetière, 21°11·6′S, 55°37·5′E, 2050 m, mixed forest with mature Acacia heterophylla, on Acacia, 2008, E. Sérusiaux s. n. (LG), P. van den Boom 39979, 40001 (h); Piton de la Fournaise, 0·5 km NW of Gîte du Volcan, 21°12·82′S, 55°41·18′E, 2040 m, Acacia heterophylla wood on N-slope, 2008, M. Brand 58353 (h).
Micarea lignaria (Ach.) Hedl.
Thallus consisting of small (100 µm across), convex, light grey granules over an algal layer growing on saxicolous mosses. Photobiont micareoid, cells round, c. 6·5–8·0 µm.
Apothecia c. 0·25 mm broad, convex, black, immarginate. Hymenium c. 60 µm, epihymenium blue. Ascospores 8 per ascus, fusiform, c. 20·0×5·2 µm, 5–7-septate.
Chemistry
Pigment in apothecia belonging to Cinereorufa-green.
Habitat and distribution
On steep rock face at 2450 m.
Notes
This collection is very small, but all characters match those of European collections. The thallus is P+ red, but is too small for chemical analysis; the shape of crystals in the thallus is identical to that of argopsin, the lichen substance diagnostic of M. lignaria. The ascospores are rather small but fall within the range of European material. The species is widespread as, according to Coppins (Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009), it occurs in Europe, North and South America, and Asia (Siberia).
Specimen examined. Réunion: Cilaos, path Le Bloc to cabane Dufour, Côteau de Kervéguen, 21°6·72′S, 55°29·70′E, 2450 m, basalt of W-exposed rock face, 2008, M. Brand 59904 (h).
Micarea melanoprasina Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807678
Thallus to 5 cm, pale green on black prothallus; apothecia 0·2–0·5 mm, dark grey to black; ascospores oval-oblong to slightly clavate, 8·5–10·5×2·8–4·0 µm, 0–1-septate; production of unkown compound.
Type: Réunion, ‘Réserve naturelle de la Roche Ecrite’, track to the summit, 20°58′6″S, 55°26′26″E, c. 1500 m, montane forest, on Acacia heterophylla, 4 November 2009, N. Magain & E. Sérusiaux s. n. (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).

Fig. 3. Photographs of the new species of Micarea described from Réunion. A & B, M. melanoprasina sp. nov. (holotype); C, M. pseudolignaria sp. nov. (holotype); D, M. sublithinella sp. nov. (holotype); E, M. takamakae sp. nov. (holotype); F, M. tenuispora sp. nov. (holotype). Scale=1 mm. In colour online.
Thallus to 5 cm across, pale green, made of coralloid goniocysts growing on bluish black prothallus; thallus surrounded by bluish black prothallus, consisting of c. 2 µm thick blue-black, irregularly intricated hyphae; goniocysts 14–30 µm, round to ovate, fused to form branched coralloid structures to 200 µm high, composed of conglutinated hyphae in a gelatinized matrix and micareoid photobiont (cells c. 4–5 µm), inspersed with small crystals (melting in boiling water forming oily drops, recrystallizing after cooling).
Apothecia 0·2–0·5 mm diam., grey to black, adnate, without visible margin, convex. Excipulum colourless or dark blue, formed by radiating hyphae. Hypothecium pale or more or less blue-aeruginose, and brownish (K+ purplish brown) in lower part. Hymenium 33–37 µm high, more or less aeruginose near base; epihymenium colourless to aeruginose. Paraphyses 1·1–1·5 µm thick, branched and anastomosing. Asci c. 32–35×12–13 µm; tholus of the Micarea-type. Ascospores 8 per ascus, oval-oblong to slightly clavate, 8·5–10·5× 2·8–4·0 µm, 0–1-septate, septum not in middle of the spore but somewhat to the wider end (old spores occasionally 3-septate).
Micropycnidia occasionally present, small (15–30 µm), immersed, colourless; microconidia shortly bacilliform, 4·0–4·5×1·0 µm.
Chemistry
Unknown 1 in thallus (K−, P−, C−). Pigment in thallus and apothecia belonging to Cinereorufa-green.
Etymology
The name of this new species refers to the bluish black prothallus that makes it very characteristic amongst species of the M. prasina group, to which it belongs.
Habitat and distribution
On trunks of trees, including Acacia heterophylla, and on branches of shrubs (Gaertnera vaginata, Chassalia coralloides, Dombeya sp.) in montane rainforest (1400–1900 m).
Notes
A distinctive species, easily recognized because of its dark prothallus and finely coralloid-goniocystose thallus, very much akin to that of M. prasina. Micarea melanoprasina is indeed related to the M. prasina group and can be distinguished by the presence of Cinereorufa-green in the thallus and apothecia, as well as the unnamed brown pigment in the apothecia. Micarea subviridescens (Nyl.) Hedl. has a dark bluish green thallus, made of dense aggregated goniocysts, dark apothecia, larger ascospores (10–18×4–6 µm) and contains prasinic acid (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009).
Additional specimens examined. Réunion: Bélouve forest, 21°03·93′S, 55°32·93′E, 1600 m, pristine montane forest, on tree, 27 v 2008, E. Sérusiaux s. n. (LG); Bois de sans Souci, 21°1·23′S, 55°22·10′E, 1350 m, montane forest, on Gaertnera vaginata, 2008, M. Brand 59994 (h); 21°01·22′S, 55°22·23′E, 1380 m, open shrub woodland, degraded montane forest, on Chassalia corallioides, 2008, M. Brand 60103 (h); E of Le Tampon, Forêt de Notre-Dame de la Paix, 21°15·1′S, 55°36·5′E, 1720 m, montane forest, on Dombeya, 2008, P. van den Boom 40775 (h); Cirque de Cilaos, Forêt du Grand Matarum, along path to Cabane Dufour, 21°07·04′S, 55°29·23′E, 1870 m, montane forest, on Dombeya, 2008, M. Brand 59740 (h).
Micarea peliocarpa (Anzi) Coppins & R. Sant.
Thallus rather small, up to 1·5 cm across, pale greenish, uneven, c. 50 µm high. Upper part c. 15 µm without algae, of loosely interwoven hyphae. Photobiont micareoid, photobiont layer inspersed with crystals (gyrophoric acid).
Apothecia up to 0·6 mm diam., whitish or dark bluish grey, often patchily coloured, with pale margin, flat or low convex, almost globose, adnate with constricted base. Outer parts of excipulum pale or bluish grey, pale inside. Hypothecium colourless. Hymenium c. 40–60 µm high; epihymenium colourless or patchily blue-grey, if pigmented then pigment also descending in hymenium. Paraphyses 1·1–1·4 µm thick, branched and anastomosing. Asci clavate, c. 35–55×12–15 µm, tholus of the Micarea-type. Ascospores 8 per ascus, fusiform with rounded ends, 3-septate, 13·6–17·0×3·6–4·7 µm.
Macropycnidia not rare, globose, c. 120–150 µm, top blue-grey or not; macroconidia curved (but not spirally), 45–58×1·1–1·3 µm, indistinctly 3-septate. Micropycnidia rare, inconspicuous, semi-superficial, c. 45 µm, pale. Microconidia narrowly fusiform with tapering ends, 6·3–8·1×0·7–0·9 µm.
Chemistry
Gyrophoric and methylhiascic acids (K−, P−, C+ red), mainly in thallus, hymenium, excipulum and pycnidia. Pigment in thallus and apothecia belonging to Cinereorufa-green.
Habitat and distribution
Mostly on soft bark of shaded Acacia heterophylla trees, at 1500–2000 m; also found on Erica, and sometimes overgrowing mosses.
Notes
Within the speciose Micarea peliocarpa group in Réunion, we recognize M. borbonica sp. nov. with 7-septate fusiform ascospores, and thus close to M. cinerea (see further comments under M. borbonica), M. boryana sp. nov. with 3-septate narrowly clavate ascospores (see under that species) and also M. peliocarpa s. str. which encompasses collections with consistently 3-septate, fusiform ascospores. The size of ascospores falls in the range of European populations as provided by Coppins [Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009: (11–)15–23(–24)×3–5(–6) µm]. However, in the Réunion populations the ascospores are on average wider than in Europe: 13·6–17·0×4·1–4·7 µm in Réunion vs. 12·2–22·5×3·5–4·2 µm in Europe (unpublished results, based on studies of 35 collections).
Specimens examined. Réunion: St-Paul, road to Piton Maïdo, 21°03·24′S, 55°21·39′E, 1500 m, edge of open woodland, on large Acacia heterophylla, 2008, M. Brand 59488 (h); Cilaos, Plateau du Petit Matarum, path from Le Bloc to Cabane Dufour, 21°06·96′S, 55°29·22′E, 1900 m, montane forest, on Acacia heterophylla, 2008, M. Brand 59756 (h); ibid., 21°07·3′S, 55°29·2′E, 1420 m, montane forest, on tree, 2008, P. van den Boom 40282 (h); Piton de la Fournaise, 0·5 km NW of Gîte du Volcan, 21°12·8′S, 55°41·18′E, 2040 m, on Acacia heterophylla in wood on N-slope, 2008, M. Brand 58356a, 58356b (h); Forêt de Bébour, path Bélouve to cabane Dufour, 3 km from Gîte Bélouve, 21°05·10′S, 55°31·36′E, 2040 m, wet high ericoid thickets, on Erica, 2008, M. Brand 58864 (h).
Micarea prasina Fr.
Thallus diffuse, c. 1 cm across, green, thin (50 µm), granular, made of goniocysts 30–40 µm diam., round, dark greyish green; goniocysts composed of thin hyphae inspersed with small crystals (melting in boiling water). Photobiont micareoid, cells c. 4–6 µm.
Apothecia up to 0.3 mm diam., dark grey to dull black, convex, adnate with narrow base, without any visible margin. Excipulum in lower part of apothecia c. 40 µm wide, formed of radiating hyphae. Hypothecium dark grey-brown (K+ violet). Hymenium to 50 µm high. Paraphyses c. 1·2–1·3 µm thick, branched and anastomosing. Asci c. 40–42×9–12 µm, tholus of the Micarea-type. Ascospores 8 per ascus, ovoid-ellipsoid, 9·3–12·8×3·8–4·3 µm, (0–)1-septate.
Micropycnidia sparse, white or grey; microconidia 6·3–7·1×0·9–1·1 µm, narrow fusiform, straight or slightly curved.
Chemistry
Micareic acid. With Sedifolia-grey pigment in inner excipulum, hypothecium and in diffuse streaks in hymenium.
Habitat and distribution
On bark of Acacia heterophylla, intermixed with the related M. levicula, and so far only found in natural stands of Acacia heterophylla near the Piton de la Fournaise.
Notes
Our material differs from Micarea prasina, as it is now circumscribed in Europe (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009), by the location of the K+ violet pigment in the hypothecium instead of the epihymenium. Otherwise the thallus made of finely dissected and coralloid goniocysts, and the production of micareic acid, are diagnostic for M. prasina. We refrain from describing a new species on that sole basis. A K+ violet hypothecium is otherwise only known from M. endoviolascens Coppins from South Africa (Coppins Reference Coppins1999), easily distinguished by its corticated squamules, and M. hypoviolascens Czarnota & Coppins from a single locality in W Scotland (Czarnota & Coppins Reference Czarnota and Coppins2005), distinguished by its areolate thallus and production of an unknown substance.
Specimens examined. Réunion: NNW of Piton de la Fournaise, trail along Ravine Savane Cimetière, 21°11·6′S, 55°37·5′E, 2050 m, mixed forest with mature Acacia heterophylla, on Acacia, 2008, P. van den Boom 39976 (h); Piton de la Fournaise, c. 0·5 km NW of Gîte du Volcan, 21°12·82′S, 55°41·18′E, 2040 m, on Acacia heterophylla in wood on N-slope, 2008, M. Brand 58355 (h).
Micarea pseudocoppinsii Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807679
Thallus diffuse, small, pale greenish, with convex soralia; apothecia to 0·4 mm, white, pale orange or partly bluish grey, convex; ascospores bacillar-ellipsoid, 11·5–13·0× 4·2–5·7 µm, (0–)3-septate; production of gyrophoric acid.
Type: Réunion, NNW of Piton de la Fournaise, trail along Ravine Savane Cimetière, 21°12·06′S, 55°41·46′E, 2050 m, mixed forest with mature Acacia heterophylla, on Acacia, 28 May 2008, P. van den Boom 39991 (LG—holotype; hb. v.d. Boom, hb. Brand 61466—isotype).
(Fig. 2H)
Thallus diffuse, small (1·5 cm across) pale greenish, made of small granules or goniocysts, locally with soralia; goniocysts 18–48 µm diam., round to ovoid, partly coalescing but not coralloid. Photobiont micareoid, c. 5–8 µm. Soralia sometimes numerous, round, convex, 0·3–0·6 mm broad; soredia light green, very fine, 11–32 µm diam.
Apothecia up to 0·4 mm diam., white, pale orange or partly bluish grey, convex, adnate with constricted base, without any visible margin. Excipulum formed by radiating, conglutinate hyphae, outer parts pale or bluish grey, pale inside, partly incrusted with gyrophoric acid crystals. Hypothecium colourless. Hymenium c. 50–55 µm high; epihymenium colourless or blue-grey in patches. Paraphyses 1·7–1·9 µm thick, simple or branched, strongly conglutinate. Asci clavate, for example 32–47×11–16 µm, tholus of the Micarea-type. Ascospores 8 per ascus, bacillar-ellipsoid, 11·5–14·7×4·2–5·7 µm, (0–)3-septate, straight to curved.
Pycnidia not found.
Chemistry
Gyrophoric acid (K−, P−, C+ red), mainly in thallus and soredia, sometimes in excipulum. Pigment in apothecia belonging to Cinereorufa-green.
Etymology
The name refers to another species of Micarea, M. coppinsii, which was dedicated to our talented colleague and friend, Dr Brian J. Coppins.
Habitat and distribution
On soft bark of Acacia heterophylla or overgrowing mosses, between 1500–2050 m elevation.
Notes
Genuine soralia are rare in Micarea. Beside M. pseudocoppinsii, they are only known in two other species: M. alectorialica, here described as new and easily identified by its black apothecia and production of alectorialic acid, and M. coppinsii Tønsberg, also producing gyrophoric acid. The latter species differs in producing methylhiascic acid together with gyrophoric acid, and has fusiform, sometimes slightly clavate, longer ascospores (20–28×4–5 µm)(Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009). Micarea viridileprosa Coppins & van den Boom has a bright, green, leproid thallus without clearly delimited soralia and has smaller ascospores [8–12(–14)×2·5–4·0 µm]. Micarea pseudocoppinsii is most likely related to the group of M. peliocarpa as it has the same type of apothecia, and to M. peliocarpa which differs by its less-developed thallus and lack of soralia.
Additional specimen examined. Réunion: Forêt de Bébour, near Gîte de Bélouve, 21°03·74′S, 55°32·31′E, 1520 m, old plantations of Acacia heterophylla trees, on Acacia, 2008, P. van den Boom 39861(h).
Micarea pseudolignaria Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807680
Thallus muscicolous, areolate, greyish green; apothecia to 0·6 mm, shining black, convex; ascospores ellipsoid, 8·0–9·2×3·0–3·3 µm, (0–)1-septate; production of unknown substance.
Type: Réunion, Forêt de Bébour, path Bélouve to cabane Dufour, 21°04·77′S, 55°31·64′E, 1740 m, soil in ericoid thickets, 2 June 2008, M. Brand 58846 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 3C)
Thallus c. 2 cm across, overgrowing decaying bryophytes, uneven, made of coalescent, 0·2–0·3 mm diam., slightly convex, greyish greenish areolae. Photobiont micareoid, cells c. 5 µm. Cortex absent but areolae partly covered by a hyaline epinecral layer. Thallus without crystals, except for lower part of photobiont layer more or less opaque due to oily drops and tiny crystals.
Apothecia up to 0·6 mm diam., shining black, convex without any distinct margin, often coalescing to form compound apothecia. Excipulum with radiating conglutinated hyphae, outer parts brownish to bluish, colourless inside. Hypothecium colourless. Hymenium c. 40 µm high, lower part more or less inspersed; epihymenium blue. Paraphyses 1·5 µm thick, strongly branched and anastomosing. Asci clavate, c. 35×9 µm, tholus of the Micarea-type. Ascospores 8 per ascus, ellipsoid, 8·0–9·2×3·0–3·3 µm, (0–)1-septate, wall c. 0·3 µm thick.
Micropycnidia superficial (i.e. on thin thallus overgrowing leafy liverworts), c. 75 µm diam., wall dark from bluish pigment. Microconidia bacillar, 4·6–5·8×0·9–1·0 µm.
Chemistry
Unknown 3 in thallus (K−, P−, C−), most probably present as tiny oil droplets or crystals. Pigment in apothecia belonging to Cinereorufa-green.
Etymology
The name refers to the widespread M. lignaria, which could be easily confused with this new species.
Habitat and distribution
Only known from the type locality. On soil, among rich stands of Cladonia, in montane ericoid shrubs.
Notes
This new species looks very much like M. lignaria (Ach.) Hedl., also found in a single locality in Réunion, but has smaller 1-septate ascospores and a different chemistry. In M. lignaria, ascospores are 3–7-septate, 16–28(–38)×4·3–5·7(–7·0) µm, and the thallus produces argopsin (var. lignaria) or unidentified xanthones (var. endoleuca). Furthermore, M. lignaria has a greenish hue in the lower part of the hymenium, which is never inspersed. The rare M. ternaria (Nyl.) Vězda, known only from the Northern Hemisphere, has a similar thallus and apothecia but differs by the lack of lichen substances and (1–)3-septate, 13–22× 3·5–5·0 µm ascospores (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009).
Micarea sublithinella Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807681
Thallus thin, with flattened rounded areoles, green; apothecia up to 0·4 mm diam.; ascospores obovoid, 12·5–15·0×5·0–5·8 µm, 0–1-septate; production of protolichesterinic acid.
Type: Réunion, NNW of Piton de la Fournaise, trail along Ravine Savane Cimetière, 21°12·06′S, 55°41·46′E, 2050 m, mixed forest with mature Acacia heterophylla, on Acacia, 28 May 2008, P. van den Boom 39980 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 3D)
Thallus c. 2 cm across, thin (25–50 µm), green, continuous or consisting of flat rounded areoles growing over a thin, slightly shiny film over the substratum. Photobiont micareoid, cells c. 5–6 µm, thallus with a few crystals.
Apothecia numerous and small, up to 0·4 mm diam. (deformed apothecia up to 0·5 mm), dull, light brownish, broadly adnate, immarginate, convex to semiglobose. Excipulum thin (less than 10 µm). Hypothecium pale. Hymenium c. 55–75 µm high; epihymenium colourless or faintly brownish, with crystals in vertical streaks. Paraphyses 1·0 µm thick, branched. Asci c. 50–70×15–16 µm, tholus of the Micarea-type. Ascospores 8 per ascus, obovoid (apical part slightly broader), 12·5–15·0×5·0–5·8 µm, 0–1-septate, wall thin (c. 0·2 µm).
Pycnidia not found.
Chemistry
Protolichesterinic acid (K−, P−, C−), with crystals mainly detected in epihymenium, but also present in thallus.
Etymology
The name refers to M. lithinella, a widespread species that could be easily confused with this new species.
Habitat and distribution
On wood of rotting trunks, or on trees, including Acacia heterophylla, in montane forests.
Notes
Most characters of Micarea sublithinella fit the the traditional concept of the genus Micarea (sensu Coppins Reference Coppins1983), but the larger asci and production of fatty acid set it apart. In the field it can easily be confused with M. lithinella (Nyl.) Hedl., a widespread species in the Northern Hemisphere, but this species is a pioneer saxicolous species in disturbed habitats, has smaller asci (35–50×8–13 µm), smaller and simple ascospores (6·5–9·5×2·5–4·0 µm) and no compounds detected (Coppins Reference Coppins, Smith, Aptroot, Coppins, Fletcher, Gilbert, James and Wolseley2009). Species related to M. lithinella, such as M. farinosa Coppins & Aptroot, also lack compounds.
Additional specimens examined. Réunion: same locality as the type, E. Sérusiaux s. n. (LG); Forêt de Bébour, trail to Takamaka, 21°05·9′S, 55°34·3′E, 1370 m, wet montane forest, on trees, 2008, P. van den Boom 40872 (h); E of Le Tampon, Forêt de Notre-Dame de la Paix, 21°15·1′S, 55°36·5′E, 1720 m, montane forest, on tree, 2008, P. van den Boom 40745 (h).
Micarea takamakae Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807682
Thallus semi-endoxylic, made of goniocysts, whitish; apothecia up to 0·2 mm diam., adnate; ascospores ellipsoid, 8·8–10·2×3·1–3·8 µm, 0–1-septate; production of unknown substance.
Type: La Réunion, WSW of St-Benoît, along the road to Takamaka, c. 3 km SW of Abondance les Hauts, 21°03·5′S, 55°38·5′E, 620 m, disturbed area with planted trees, on decaying tree, 11 June 2008, P. van den Boom 41009 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 3E)
Thallus inconspicuous, diffuse, partly endoxylic on soft, decaying wood, whitish, made of dispersed, round goniocysts, c. 30–75 µm diam., either in empty dead cells of the bark or, when superficial, goniocysts covered by clear necrotic tissue. Photobiont micareoid, cells c. 5–7 µm. Crystals detected in goniocysts.
Apothecia very numerous and small (up to 0·2 mm diam.), black, adnate with constricted base to nearly stipitate, first flat without any distinct margin, then convex, often forming clusters. Excipulum well developed in under part, made of chondroid tissue; outer part hyaline, inner part locally brown. Hypothecium pale, opaque because of numerous small crystals. Subhymenium reddish brown. Hymenium c. 35 µm high; epihymenium dull blue-green and brownish, with small dark granules in K. Paraphyses 1·4 µm thick, branched. Asci c. 27–30×11–13 µm, tholus of the Micarea-type. Ascospores 8 per ascus, ellipsoid, 8·8–10·2×3·1–3·8 µm, 0–1-septate, wall c. 0·3 µm thick.
Pycnidia not found.
Chemistry
Unknown 2, assumed to be confluentic acid or a related compound (K−, P−, C−). Pigment in epihymenium belonging to Cinereorufa-green.
Etymology
The name refers to a spot on the northern flank of the island, ‘Le Cassé de Takamaka’, a landscape of dramatic beauty.
Habitat and distribution
Only known from the type locality, at rather low elevation (620 m) in a high rainfall area, on decaying wood of standing trunk.
Notes
Micarea takamakae has an excipulum and hymenium very much akin to those of M. erratica, but all other characters are quite different and set it apart from that species. Confluentic acid has never been reported in the genus but all chemical features point to that substance. Small ascospores are a further interesting feature of this new species, amongst the species found on Réunion and not belonging to the M. micrococca-prasina aggregate.
Micarea tenuispora Brand, van den Boom & Sérus. sp. nov.
MycoBank No.: MB807683
Thallus very thin to endophloeodal, isidiate or made of isidiiform areolae, isidia small cylindrical to coralloid, up to 0·3 mm high; apothecia to 0·9 mm, dark grey to black, ascospores acicular, narrowly clavate, 31–35× 2·2–2·5 µm, 3–5-septate; production of gyrophoric acid.
Type: Réunion, Cirque de Cilaos, Forêt du Grand Matarum, trail to Caverne Dufour, 21°07·3′S, 55°29·2′E, 1420 m, montane forest, on Cryptomeria, 31 May 2008, P. van den Boom 40231 (LG—holotype; hb. v.d. Boom, hb. Brand—isotype).
(Fig. 3F)
Thallus to 5 cm across, pale greenish to brownish, discontinuous, thin (c. 50 µm thick), consisting of scattered areolae, soon forming isidia or becoming isidiiform, on thin endo- to epiphloeodal dark bluish hypothallus with narrow, colourless or locally dark brownish or bluish hyphae; mature thallus always with isidiiform areolae (c. 0·2 mm high, 0·05–0·07 mm thick). Cortex absent, often with thin epinecral layer. Photobiont micareoid. Thallus filled with crystals of gyrophoric acid.
Apothecia up to 0·8 mm diam., sessile to substipitate, black, usually with a pale margin when young, first flat, without distinct rim, then usually but not always semiglobose. Excipulum with radiating hyphae, varying in colour (often in the same apothecium) from pale (then with crystals) to superficially bluish and centrally light brown. Hypothecium pale brown to dark bluish. Hymenium c. 55 µm high; epihymenium greyish bluish in patches. Paraphyses c. 1·3 µm thick, branched and anastomosing. Asci c. 44×11 µm, tholus of the Micarea-type. Ascospores 8 per ascus, in one bundle, sometimes twisted, acicular, apically inflated and tapering towards the base, 31–35×2·2–2·5 µm, 3–5-septate.
Micropycnidia scarce, inconspicuous, sessile, c. 110 µm, with blue-green top, brownish wall. Microconidia narrow fusiform, average 3·8–4·1×1·1–1·2 µm.
Chemistry
Gyrophoric acid (K−, P−, C+ red), mainly in thallus, but also in pale parts of apothecia and in pycnidia. Pigment in thallus and apothecia belonging to Cinereorufa-green. Brown pigment in hypothecium K+ reddish to purple.
Etymology
The name was chosen to emphasize the thin ascospores of this species.
Habitat and distribution
On bark of the exotic Cryptomeria japonica, between 1200–1500 m elevation, at edges of disturbed natural forests.
Notes
Micarea tenuispora is easily distinguished by its ‘isidiate’ thallus and 3–5-septate ascospores. Isidia or isidiiform thallus areolae are reported in only two species of Micarea, both described here as new. For differences to M. isidiosa sp. nov., see under that species.
Additional specimen examined. Réunion: Cilaos, Forêt du Grand Matarum, 21°07·17′S, 55°29·10′E, 1450 m, disturbed margin of montane forest, on Cryptomeria, 2008, M. Brand 58685 (h).
We are greatly honoured to publish our paper in this special issue of The Lichenologist for the 65th birthday of our most distinguished colleague and friend, and the world expert on Micarea taxonomy, Dr Brian J. Coppins.
Field studies in Réunion were made possible with the help and advice from the Parc National de La Réunion, especially through the courtesy of Mr B. Lequette and Mr J. M. Pausé. Dr Cl. Ah-Peng and Prof. D. Strasberg of the University of La Réunion in Saint-Denis were also very helpful. We thank them all most sincerely. We further thank the Senior Editor of The Lichenologist, Prof. P. D. Crittenden, and the three Editors of this special volume (Dr Alan Fryday, Dr Gintaras Kantvilas and Dr Chris Ellis). Both referees contributed to the final version of this manuscript as their notes and suggestions were very helpful: we thank them warmly.







