Adherence is regarded as one of the main factors for a successful treatment of many diseases (Reference Gorenoi, Schoenermark and Hagen1). The World Health Organization (WHO) estimates an adherence rate of 50 percent for long-term therapies for chronic conditions in developed countries (Reference Sabaté2). Nonadherence can result in suboptimal treatment outcomes and adverse events. Consequently, enormous costs are caused for healthcare systems (Reference Gorenoi, Schoenermark and Hagen1). Adherence is a particularly crucial aspect with respect to highly active antiretroviral therapy (HAART) treatment outcomes and avoiding of drug resistant strains (Reference Thompson, Mugavero and Amico3). However, many HIV-infected patients show low adherence due to their frequently difficult living conditions and the partly complex intake regimen. Numerous different interventions aim to improve patient adherence. Adherence-improving interventions contain educative, behavioral, or psychosocial elements as well as combinations of these. Furthermore, a simplification of the intake regimen is likely to affect adherence. The Directly Observed Therapy (DOT), which involves dispensing pharmaceutical treatment in the presence of a healthcare professional is an option for patients in difficult life situations, for example, intravenous drug users in maintenance organizations or prisoners (Reference Scalera, Bayoumi, Oh, Risebrough, Shear and Tseng4). A significant influence on patient adherence and on the viral load has been demonstrated for part of these interventions (Reference Haynes, Ackloo, Sahota, McDonald Heather and Yao5). Previous studies have also shown that adherence can be associated with quality of life dimensions in patients receiving HAART (Reference Carballo, Cadarso-Suarez and Carrera6). Furthermore, evidence already exists for some indications showing that adherence interventions can reduce healthcare resource usage, and, therefore, also costs (Reference Carballo, Cadarso-Suarez and Carrera6;Reference Golay7).
The objective of this systematic review was to analyze the cost-effectiveness of different adherence interventions as well as influencing factors for HAART in HIV-infected patients in developed countries (WHO stratum A).
METHODS
Based on the study question a broad search strategy was developed to reach maximum sensitivity. The following databases were searched without limiting the search period (conduct of search on September 27th, 2011): EMBASE (by means of Embase), MEDLINE (by means of Embase), NHS Economic Evaluation Database (by means of Cochrane library), CINAHL (by means of Ebsco), HEED (by means of Wiley), and EconLit (by means of Ebsco). The search strategy was adapted for each database and combined diverse expressions including medical subject headings, synonyms, and acronyms for the topic adherence and HIV. The search filter for economic studies provided by the Scottish Intercollegiate Guidelines Network and Emtrees for HIV and adherence was used in the EMBASE search. The full search strategies for the individual databases can be found in Supplementary Material 1, which can be viewed online at www.journals.cambridge.org/thc2013093. The references of all included studies were cross-checked as well. Inclusion criteria were as follows: (i) adult patients (≥18) with HIV infection, (ii) current treatment with HAART, (iii) interventions to increase adherence compared with standard of care or other adherence enhancing interventions, (iv) incremental cost-effectiveness or cost-utility analysis, (v) located in countries classified into WHO-stratum A, (vi) articles published in English or German, and (vii) obtainable as full-text article
Comparisons of different application forms or dosage variations were excluded as the effectiveness cannot be definitely linked with the adherence intervention (e.g., simplification of intake), but rather other drug reactions like adverse events.
The abstracts of all hits and, in case of relevance, the entire articles were screened and selected according to the predefined inclusion and exclusion criteria described above. The study quality of the included articles was evaluated by using the instrument for assessing economic studies provided by the Ludwig Boltzmann Institute (LBI) in Austria (8). This instrument consists of eleven dimensions and a total of fifty-eight items to evaluate the validity and the formal quality of economic evaluations. The dimensions comprise the study question, the evaluation frame, the method of analysis and modeling. Health effects, costs, discounting, the presentation of results, the handling of uncertainty, equity, the discussion and conclusion are addressed in addition. This instrument has been applied for HTAs (Reference Zechmeister and Wild9) and allows a comprehensive and detailed assessment of the study quality (Reference Dreier, Borutta, Stahmeyer, Krauth and Walter10). Study selection and evaluation were independently performed by two reviewers.
Study characteristics and results were subsequently extracted in standardized tables by one reviewer and checked by a second for quality assurance.
Any differences in study selection, evaluation, and extraction were solved in discussion or by involving a third opinion. A quantitative data synthesis was not planned due to the expected study heterogeneity, especially with regard to adherence interventions, included patients and study methods, but was not definitely excluded beforehand.
In view of the many different parameter variations for sensitivity analyses, it was decided to state only the range of results and the variations that resulted in an exceedance of a threshold of USD 50,000/QALY (11).
RESULTS
Sixteen of a total of 959 studies (after the technical removal of duplicates) identified by the search strategy, seemed potentially relevant and were read in full text. Of these, twelve studies did not meet the predefined inclusion criteria and were, therefore, excluded from analysis. In seven cases, an adherence intervention was not the object of analysis. Three were excluded because they did not determine the incremental cost-effectiveness ratio of different interventions (or no intervention). For a list of excluded studies see Supplementary Material 2, which can be viewed online at www.journals.cambridge.org/thc2013094. Two studies were no full-text articles. In the end, four studies were eligible for this review (Reference Braithwaite, Roberts and Justice12–Reference Zaric, Bayoumi, Brandeau and Owens15). Figure 1 (flow-chart) illustrates the selection process.

Figure 1. Flow-chart of study selection.
The study by Braithwaite et al. (Reference Braithwaite, Roberts and Justice12) investigated the influence of the quality of evidence on the results in economic evaluations as a methodological question. The cost-effectiveness of adherence interventions for HAART is used here as a basis to compare the impact of varying study quality on the results in health economic modeling approaches. Consequently, almost none of the evaluation items were stated, even though the study fulfilled all inclusion criteria. All remaining three studies showed various methodological flaws. The full evaluations of the three studies are available in Supplementary Material 3, which can be viewed online at www.journals.cambridge.org/thc2013095. In particular descriptions of resource use and underlying costs were neither transparent and traceable nor fully reported in any of the three studies. Furthermore, the determination of QALYs was not reported. Two studies compared only two alternatives of many possible interventions and the reason for the selection is not stated (Reference Freedberg, Hirschhorn and Schackman13;Reference Zaric, Bayoumi, Brandeau and Owens15). None of the studies considered equity aspects. Another deficit is that discounting of the health status was not carried out and reasons for this omission were not given, neither by Freedberg et al. (Reference Freedberg, Hirschhorn and Schackman13) nor by McCabe et al. (Reference McCabe, Goldie and Fisman14). The most serious deficits were revealed in the study by McCabe et al. (Reference McCabe, Goldie and Fisman14) while Zaric et al. (Reference Zaric, Bayoumi, Brandeau and Owens15) showed the least deficits.
Two of the included studies consider DOT versus standard of care or self-administration (Reference Braithwaite, Roberts and Justice12;Reference McCabe, Goldie and Fisman14). The other two deal with educational adherence interventions versus standard care (Reference Freedberg, Hirschhorn and Schackman13;Reference Zaric, Bayoumi, Brandeau and Owens15). According to the study by Freedberg et al. (Reference Freedberg, Hirschhorn and Schackman13) standard care already includes some components to increase adherence such as nurse education, education material and reminder devices. The primary outcome and outcome for incremental analysis in all studies are QALYs. McCabe et al. (Reference McCabe, Goldie and Fisman14) focus on DOT for pregnant women (Reference McCabe, Goldie and Fisman14) and Zaric et al. (Reference Zaric, Bayoumi, Brandeau and Owens15) on counseling for men who have sex with men (MSM). In most studies, the observed region is the United States (Reference Freedberg, Hirschhorn and Schackman13–Reference Zaric, Bayoumi, Brandeau and Owens15). The studies in which the setting is reported are conducted in an outpatient context (Reference Freedberg, Hirschhorn and Schackman13;Reference McCabe, Goldie and Fisman14). Modeling refers to a life time period (Reference Freedberg, Hirschhorn and Schackman13;Reference McCabe, Goldie and Fisman14) or 20 years (Reference Zaric, Bayoumi, Brandeau and Owens15). Only direct healthcare costs are considered (Reference McCabe, Goldie and Fisman14;Reference Zaric, Bayoumi, Brandeau and Owens15), except for time costs for patients and healthcare professionals (Reference Freedberg, Hirschhorn and Schackman13). Detailed study methods and the design of the individual studies are presented in Table 1.
Table 1. Methods/Design of Included Studies
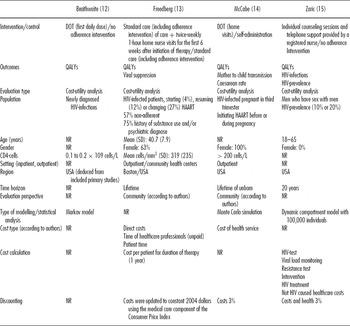
NR, Not reported; SD, standard deviation.
In the study that compared DOT with no intervention (based on studies with the highest quality) a cost-utility ratio of USD 158,000/QALY was calculated (Reference Braithwaite, Roberts and Justice12). A sensitivity analysis for clinical and cost data was not performed due to the focus on the methodological question. Freedberg et al. (Reference Freedberg, Hirschhorn and Schackman13) estimated that twice weekly home nurse visits for the first 6 weeks after changes in the HAART regimen were highly cost-effective (USD 14,100/QALY). The sensitivity analysis confirmed the robustness of the result. All cases were cost-effective whereby a USD 24,200/QALY was not exceeded. DOT for HIV-infected pregnant women was cost-effective, too (Reference McCabe, Goldie and Fisman14). The cost-utility ratio in this study was USD 14,233/QALY. However, baseline viral load reductions, a strong decrease of viral load reduction (base case scenario 0.8 Log10, sensitivity analysis 0.5 Log10), by HAART and an increase of intervention costs (HAART and DOT, values not quantified) produced cost-utility ratios above USD 50,000/QALY up to USD 630,000/QALY. Zaric et al. (Reference Zaric, Bayoumi, Brandeau and Owens15) also found that counseling for MSM was highly cost-effective (USD 7,392/QALY). Only a strong increase of intervention costs (base case scenario USD 21.73 per counseling session and USD 5.42 per month for telephone support; sensitivity analysis USD 200 per counseling session and USD 100 per month for telephone support) resulted in a small exceedance of USD 4,300/QALY of the cost-effectiveness threshold. A stochastic sensitivity analysis in this study predicted a chance of 89 percent for the intervention to be cost-effective.
Study results are shown in Table 2 (for more details see appendix 4). The nonincremental data for Zaric et al. refer to 100,000 people because also noninfected subjects were included in the modeling (Reference Zaric, Bayoumi, Brandeau and Owens15).
Table 2. Results of Included Studies

aAll non-incremental data per 100,000 persons.
bBased on highest quality study.
NR, Not reported.
DISCUSSION
The aim of this review was to evaluate the cost-effectiveness of adherence interventions for HAART in HIV-infected patients. Four cost-utility studies that met the inclusion criteria were identified. The authors identified studies examining DOT for newly HIV-infected and HIV-infected pregnant women initiating HAART and education interventions for MSM and predominately patients with substance use problems and/or psychic disorders. Study methods varied widely, too. Overall, the study quality was poor, that is, each study has some serious methodological flaws. In three out of four studies, the base case scenarios showed cost-effective results (Reference Freedberg, Hirschhorn and Schackman13–Reference Zaric, Bayoumi, Brandeau and Owens15). In addition, in one study most scenarios (Reference McCabe, Goldie and Fisman14) and in two studies (Reference Freedberg, Hirschhorn and Schackman13;Reference Zaric, Bayoumi, Brandeau and Owens15) all cases of sensitivity analyses were cost-effective.
For the study that examined nurse home visits in addition to standard adherence interventions and the study that examined DOT for pregnant women in cost-utility-analyses, the costs per QALY in the base-case scenario were each approximately USD 14,000 (Reference Freedberg, Hirschhorn and Schackman13;Reference McCabe, Goldie and Fisman14). In the study that evaluated counseling and telephone support by a nurse the incremental corresponding base case cost-utility ratio was USD 7,392 (case of 10 percent baseline prevalence), respectively, USD 8,682 (case of 20 percent baseline prevalence) (Reference Zaric, Bayoumi, Brandeau and Owens15). Also the univariate and bivariate sensitivity analyses of these three studies with a presumed maximum threshold of USD 50,000/QALY (11) showed predominantly cost-effective results for variations of costs, effectiveness parameters and adherence rates (Reference Freedberg, Hirschhorn and Schackman13–Reference Zaric, Bayoumi, Brandeau and Owens15). Exceptions were a baseline viral load less than 1,000 copies/mL, a sharp decrease of the viral load reduction and an increase of intervention costs (HAART and DOT) for pregnant women in the study by McCabe et al. (Reference McCabe, Goldie and Fisman14) as well as an increase of intervention costs for MSM in Zaric et al. (Reference Zaric, Bayoumi, Brandeau and Owens15). Furthermore, the methodological study by Braithwaite et al. (Reference Braithwaite, Roberts and Justice12) that examined DOT bases the calculation on the highest quality studies costs adding up to over USD 150,000/QALY in the base case scenario. However, an interpretation is not possible because almost all model assumptions were not reported, nor is it possible to deduce which parameters were affected by the elimination of low evidence. However, as the quality of evidence might have a strong influence on the results of the cost-effectiveness of adherence interventions the quality of the remaining studies has to be scrutinized critically. The reason is that the identification strategies and quality of studies used to determine input parameters remain unclear. The other included studies examined the interventions only in subgroups with a larger chance of cost-effectiveness. Freedberg et al. (Reference Freedberg, Hirschhorn and Schackman13) looked primarily at a population with a history of substance use problems and/or mental disorders and consequently a high rate of nonadherence. The probability of cost-effective results in such study collectives with low baseline adherence rates is higher, because low baseline adherence rates allow stronger adherence rate changes (Reference Goldie, Paltiel and Weinstein16) and consequently QALY gains. So for a typical population approximately 1.2 QALYs was gained by perfect adherence (Reference Munakata, Benner, Becker, Dezii, Hazard and Tierce17) whereas Freedberg et al. (Reference Freedberg, Hirschhorn and Schackman13) estimated a gain of 6.4 QALYs. Zaric et al. (Reference Zaric, Bayoumi, Brandeau and Owens15) included only MSM. In these populations with relatively high transmisson rates and a high prevalence (18;19), there is a greater possibility for transmisson/incidence and prevalence reductions, therefore, also for cost-effectiveness. The study by McCabe et al. (Reference McCabe, Goldie and Fisman14) considered exclusively pregnant women. Moreover, the probability of cost-effectiveness is higher in this population due to the avoided heavy life time costs for unborn children in case of a mother–child transmission which is reflected by the low value for QALYs gained.
A further limitation is that only the cost-effectiveness of educational adherence interventions and DOT are examined. Especially DOT is exclusively an option for individuals in difficult life situations because it suggests intensive disturbance of the private sphere. We did not find any evaluations dealing with other adherence interventions, like intensive reminder interventions, other educational strategies, incentives or combinations of them that also have (Reference Hart, Jeon and Ivers20) a significant effect on adherence and clinical outcomes like DOT (Reference Haynes, Ackloo, Sahota, McDonald Heather and Yao5). To our knowledge, no gold standard or standard of care exists in most countries at present, and so these are only a few out of many possible strategies to increase adherence in HIV-infected patients. The cost-effectiveness remains unknown for other interventions and in less specified populations. Patient groups that could benefit from an adherence intervention should be conclusively justified before starting the adherence intervention. In general, these are only patients with an adherence rate below the necessary adherence for virologic response (Reference Ding, Wilson, Modjarrad, McGwin, Tang and Vermund21). Like many economic evaluations in health care (Reference Jefferson and Demicheli22), each included study also revealed serious methodological deficits. If this fact is taken into account, and in particular insufficient cost data, then the validity of the study results has to be doubted, although cost-effectiveness results were calculated for most scenarios of sensitivity analyses. Methodological deficits and heterogeneity were the main reasons why a prior evidence synthesis of economic evaluations for adherence enhancing interventions did not find clear results for cost-effectiveness of other indications like asthma (Reference Elliott, Barber and Horne23).
Sensitivity analysis revealed that, at a presumed threshold of USD 50,000/QALY, the viral load and intervention costs seem to be factors influencing cost-effectiveness for DOT in HIV-infected pregnant women. This concerned only severe parameter variations in two studies for HIV-infected pregnant women and MSM (Reference McCabe, Goldie and Fisman14;Reference Zaric, Bayoumi, Brandeau and Owens15) and moderate variations in one study (Reference McCabe, Goldie and Fisman14), while in the other studies, there were no exceedances of presumed threshold; this fact also makes a general interpretation of influencing factors difficult. But the results appear to be robust even in specific populations.
Nevertheless, this systematic review suggests that adherence interventions for HIV-infected patients, especially for patients with assumed low baseline adherence, have the potential of being cost-effective. Because high adherence is a factor to reach optimal therapy outcomes and consequently, a high quality of life the cost-effectiveness of other interventions and for a more comprehensive population should be also examined. Adherence interventions for HAART receiving HIV-infected patients are not integrals part of most health systems yet although they may contribute to better care of affected patients and to avoiding resistance (Reference Cadosch, Bonhoeffer and Kouyos24). For that reason, the cost-effectiveness of existing adherence interventions should be assessed with high quality evaluations to facilitate decisions about the reimbursement of adherence interventions. Behavioral strategies like reminder devices or other educational strategies have also shown an effect on clinical outcomes and adherence and, therefore, should be taken into account for a wider application in HIV-effected persons (Reference Haynes, Ackloo, Sahota, McDonald Heather and Yao5) and for the evaluation of cost-effectiveness. Furthermore, psychoeducative interventions, for example, motivational interviewing or behavioral therapy for groups with mental problems require an examination (Reference Parsons, Golub, Rosof and Holder25).
CONCLUSION
Only evaluations of two types of adherence interventions for special study populations were identified in this review. All studies included revealed substantial methodological deficits. Thus, the cost-effectiveness of adherence interventions in general cannot be definitely assessed, nor is it possible to conclude which factors have a significant influence on the probability of being cost-effective. However, this systematic review indicates that adherence interventions for HIV-infected patients may be cost-effective under certain conditions.
Moreover, health economic evaluations for further populations and adherence interventions need to be taken into account which should be conducted based on high-quality studies and cost data.
SUPPLEMENTARY MATERIAL
Supplementary Material 1: www.journals.cambridge.org/thc2013093
Supplementary Material 2: www.journals.cambridge.org/thc2013094
Supplementary Material 3: www.journals.cambridge.org/thc2013095
CONTACT INFORMATION
Tim Mathes (Tim.Mathes@uni-wh.de), Dawid Pieper, Sunya-Lee Antoine, Dr. Michaela Eikermann, Institute for Research in Operative Medicine, Witten/Herdecke University, Cologne, Germany
CONFLICTS OF INTEREST
All authors report a payment for manuscript preparation to their institution from Janssen-Cilag.





