Introduction
Sjögren's syndrome is a rheumatological condition, first described by Henrik Sjögren in 1933.Reference Ng1 It is characterised by lymphocytic infiltration in exocrine organs, which results in systemic chronic inflammation. A typical clinical presentation encompasses the sicca symptoms of xerophthalmia, xerostomia and diffuse parotid gland swelling.
Sjögren's syndrome is classified into two general categories. Primary Sjögren's syndrome is an isolated sicca syndrome without any underlying rheumatological conditions. Secondary Sjögren's syndrome is associated with other rheumatological conditions, such as systemic lupus erythematosus, rheumatoid arthritis or scleroderma. Given the overlap of clinical presentations, it can be a challenge to diagnose Sjögren's syndrome based on clinical features alone.
Until recently, the commonly used classification and diagnostic criteria was the American–European Consensus Group criteria, proposed in 2002.Reference Vitali, Bombardieri, Jonsson, Moutsopoulos, Alexander and Carsons2,Reference Langegger, Wenger, Duftner, Dejaco, Baldissera and Moncayo3 Current criteria was further developed by the Sjögren's International Collaborative Clinical Alliance (‘SICCA’) investigators, and this is the most widely accepted criteria in both European and North American institutions at present. Diagnosis of Sjögren's syndrome according to Sjögren's International Collaborative Clinical Alliance criteria requires two of the three following findings: positive serum findings for anti-Ro/SSA and/or anti-La/SSB antibodies, an ocular staining score of 3 or greater, and lymphocytic sialadenitis on sublabial minor salivary gland biopsy.Reference Shiboski, Shiboski, Criswell, Baer, Challacombe and Lanfranchi4
Minor salivary gland biopsy is considered the definitive test to establish diagnosis of Sjögren's syndrome.Reference Daniels5 Various surgical techniques have been described, targeting harvest of minor salivary glands from the lower lip. This involves an incision made at the vascular mucosal surface of the lower lip, with minor salivary gland lobules excised for histopathological analysis. The identification of minor salivary glands is often made difficult because of bleeding from a vascular field, which obscures the salivary glands that require harvesting and puts sensory nerve fibres at risk. A dry operative field is essential to minimise morbidity, and to ensure adequate harvest of at least four to five glands for accurate histological analysis.
We describe a technique of harvesting minor salivary glands in a bloodless operative field using a chalazion ophthalmic clamp under local anaesthesia, and report our clinical outcomes.
Materials and methods
A prospective study was performed on patients of a single surgeon, who were being treated at a tertiary referral teaching hospital in South Dublin. Medical records, operative notes and histopathological results were reviewed, and data collected. Written consent was obtained for intra-operative medical photography.
All patients undergoing minor salivary gland biopsy between July 2017 and April 2018 were reviewed. Inclusion criteria were limited to include those patients who had undergone minor salivary gland biopsy utilising a chalazion clamp, with subsequent post-operative clinic attendance. Those who failed to attend the post-operative review, those with missing medical records on post-operative review and those lost to follow up, were excluded.
Operative technique
A standard 9 cm chalazion clamp with a 30 × 20 mm ellipsoid fenestra is utilised for all procedures (Figure 1a, 1b). Traction is applied at the edge of the lower lip to reflect the lower lip, and forward and downward to expose the inner lip mucosal surface. The chalazion clamp is applied over the reflected lower lip. The fenestrated leaf of the clamp rests on the mucosal surface, while the solid leaf rests against the skin surface (Figure 2a).

Fig. 1. (a) Frontal and (b) side views of a standard 9 cm chalazion clamp.
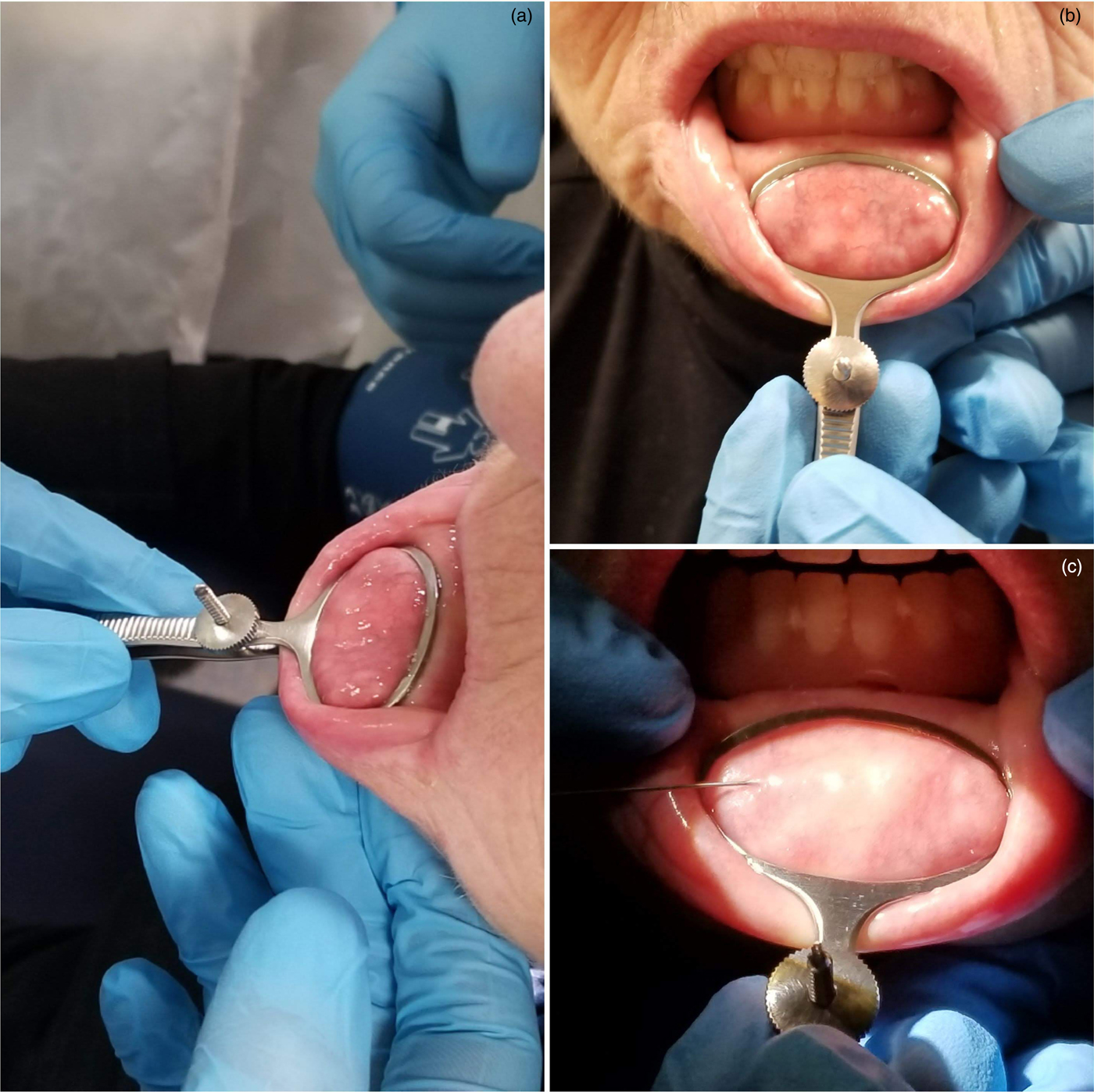
Fig. 2. Photographs showing: (a) application of chalazion clamp, (b) the clamp tightened (note the distended minor salivary glands), and (c) infiltration of a local anaesthetic agent with adrenaline.
The clamp is tightened to allow for intra-operative haemostasis, which also brings distended abnormal minor salivary glands to the mucosal surface (Figure 2b). Care is taken not to over-tighten the clamp, to avoid causing discomfort to the patient.
Using a dental syringe, 1–1.5 ml of 1 per cent lignocaine with 1:80,000 adrenaline solution is infiltrated locally into the fenestrated surgical field, to ensure adequate anaesthesia and to aid haemostasis (Figure 2c).
Once adequate anaesthesia is achieved, a 2 cm transverse incision is made, exposing submucosal tissue (Figure 3a). Enlarged abnormal minor salivary glands may readily extrude from the incision site. These are excised with sharp dissection for analysis (Figure 3b). We recommend a harvest of at least five glands for biopsy (Figure 3c). Superficial sensory nerve fibres are easily identified given the dry surgical field. Preventing injury to these nerves is paramount to avoid paraesthesia of the lip post-operatively (Figure 3d).

Fig. 3. Photographs showing: (a) mucosal incision, (b) excision of a minor salivary gland, (c) further blunt dissection, and (d) superficial sensory nerve fibre identification.
Closure is achieved via absorbable non-stained sutures, placed in an interrupted fashion, using a round-bodied needle. The sutures are left long, to prevent chafing of the lower alveolar gum mucosa (Figure 4a). The chalazion clamp is then removed and mild pressure is applied to secure haemostasis on the mucosal edges (Figure 4b).

Fig. 4. Photographs showing: (a) an absorbable interrupted suture, and (b) haemostasis secured.
Results
Twenty-three patients met the inclusion criteria, of which 21 were female and 2 male. The mean age was 50.2 years (range, 40.6–73.4 years).
The minor salivary glands were positively identified in all 23 patients. The histopathological reports of 17 patients included the specific count of glands positively harvested during the biopsy. A further five patients did not have a specific count of glands analysed, but were reported as having had multiple glands harvested. All intra-operative records specified a sample count of five glands. Forty per cent of patients had a final gland count exceeding the recorded number of glands sampled, while less than 20 per cent had a count below the intended sampling count target. The numbers of salivary glands obtained are reflected in Figure 5.

Fig. 5. Summary of the gland count obtained for each biopsy attempt, as reported on histopathology analysis. The glands were positively identified on the histopathological reports of 22 patients. There was no record of gland count for one patient. Five patients did not have a specific count of glands, but were reported as having had multiple glands harvested.
The final histopathology reports successfully identified positive findings for Sjögren's syndrome in 9 patients, while the reports for 10 patients were negative. Four patients had inconclusive findings. None of the 23 patients included in the study reported the surgical complication of lip paraesthesia at the 2-week post-operative review.
Discussion
Chalazion clamps are specially designed instruments, typically used by ophthalmic surgeons to perform incision and drainage of chalazions, or non-infective meibomitis of the tarsal plate on an eyelid. Much like the eyelids, the mucosa of the oral cavity and lips is moist, vascular, and pliant. Their use in the non-ophthalmic surgery setting was proposed by Garcia and colleagues as far back as the 1970s.Reference Garcia and Davis6
Chalazion clamps are ideal instruments for minor salivary gland biopsy. The fenestrated leaf provides adequate exposure to the operative field. The tourniquet effect to the operative field provides good haemostasis, without injuring soft tissue. The perpendicular attachment of the handle provides adequate and ergonomic lip stabilisation for the assistant, without major disruption to the operative surgeon.
Recent international consensus guidelines have highlighted the importance of minor salivary gland biopsy in diagnostic criteria for Sjögren's syndrome.Reference Shiboski, Shiboski, Criswell, Baer, Challacombe and Lanfranchi4 A systematic review on minor salivary gland biopsy by Guellec et al. concluded a high diagnostic value for Sjögren's syndrome, with 88 per cent (standard deviation (SD) ± 11.7) specificity and 78.8 per cent (SD ± 11.2) sensitivity, with a positive predictive value of 87.6 per cent (SD ± 9.5) and a negative predictive value of 79.0 per cent (SD ± 16.9).Reference Guellec, Cornec, Jousse-Joulin, Marhadour, Marcorelles and Pers7
Typical minor salivary gland lobules are spherically shaped, appearing white and semi-translucent. Other reported techniques utilising vasoconstrictive agents during preparation of the operative field invariably lead to minor haemorrhage without haemostatic pressure. This results in blood-stained tissues that make minor salivary glands hard to distinguish from surrounding lip musculature.
The most widely described immediate complication is bleeding. Haemostatic operative techniques ensure prophylactic haemostasis with clamp pressure, without the need for bipolar diathermy. Clear identification of sensory nerve fibres avoids inadvertent injury to the nerves, preventing paraesthesia, which is the commonest medium- and long-term complication reported (6–11 per cent).Reference Pijpe, Kalk, van der Wal, Vissink, Kluin and Roodenburg8,Reference Caporalli, Bonacci, Epis, Bobbio-Pallavincini, Morbini and Montecucco9
• Minor salivary gland biopsy is considered definitive for Sjögren's syndrome diagnosis
• Common complications of minor salivary gland biopsy include operative field bleeding and lip paraesthesia
• Most techniques described are non-haemostatic
• A haemostatic minor salivary gland biopsy technique using a chalazion clamp is described
• This haemostatic technique is advantageous, with respect to biopsy yield and common minor salivary gland biopsy complications
Our described technique provides a clear and bloodless operative field, and enables a superior yield compared to traditional non-haemostatic techniques described in the medical literature.
Conclusion
The use of the chalazion clamp during minor salivary gland biopsies stabilises the lip and provides a clear bloodless operative field. This technique facilitates a superior yield, ensuring adequate sampling of appropriate glands for histopathological analysis, and minimises the complications associated with traditional techniques.
Competing interests
None declared







