Urinalysis and urine culture are commonly ordered tests among hospitalized patients suspected of urinary tract infection (UTI). However, these tests are often ordered for patients without clinical suspicion of UTI, leading to unnecessary testing and increased hospital costs.Reference Trautner1–Reference Saint3 Positive urine cultures are a major driver for antibiotic treatment.Reference Hartley, Kuhn and Valley4–Reference Trautner, Bhimani and Amspoker11 Several studies have reported that the treatment of asymptomatic bacteriuria (ASB) does not affect patient outcomes and leads to unnecessary antibiotic use, increasing the prevalence of antibiotic-resistant organisms and Clostridium difficile infection.Reference Linares, Thornton, Strymish, Baker and Gupta12–Reference Gupta, Hooton and Naber14 Despite Infectious Disease Society of America and other professional societies’ recommendations to avoid antibiotic prescriptions for asymptomatic bacteriuria,Reference Gupta, Hooton and Naber14–17 its treatment is still common.
Previous interventions to prevent unnecessary urine testing have included provider education, use of pocket cards, antimicrobial stewardship efforts, reflex urine culture cancellation and 2-step urine culture ordering.Reference Pavese, Saurel and Labarere6, Reference Kelley, Aaronson, Poon, McCarter, Bato and Jankowski7, Reference Linares, Thornton, Strymish, Baker and Gupta12, Reference Trautner, Grigoryan and Petersen13, Reference Bonnal, Baune and Mion18–Reference Stagg, Lutz and Kirpalaney22 However, data on the effect of changes in electronic order sets and its role on inpatient urine testing practices are limited.
In this study, we evaluated the impact of changes to the inpatient urine orders in a computer physician order entry (CPOE) system on the urine culturing practices of a large urban, academic medical center.
Methods
Setting
This retrospective before-and-after study included patients admitted to Barnes-Jewish Hospital (BJH), a 1,250-bed teaching hospital, from January 1, 2015, to August 31, 2017, who had ≥1 urine culture ordered during their stay. Patients who were admitted during the study period but did not have a urine culture ordered during their stay and patients who had their urine cultures obtained at an outpatient settings or the emergency department (ED) were excluded.
Intervention
A staged intervention was performed to clarify test names and to reduce the number of reflex urine cultures performed for nonspecific indications (eg, isolated proteinuria) by making changes to the urine reflex test panel at BJH (Table 1). This intervention was initiated in the CPOE system on January 28, 2016. E-mail notification to providers with the new urine reflex tests was sent prior to initiation. The inclusion of the new reflex tests in commonly used order sets within the CPOE system (eg, medical intensive care unit admission orders) was completed on April 19, 2016; therefore, April 2016 was used as the intervention month. January 2015 through April 2016 was the preintervention period and May 2016 through August 2017 was the postintervention period.
Table 1. Urine Order Set Definitions During the Study Period

Note. UA, urinalysis.
a Urine Flex and Urine Flex C/S were the same orders with different names during the preintervention period;
b UA Reflex and UA W/Reflexed were the same orders with different names during the preintervention period.
c Urine Macro, Urinalysis, UA Macro and UA Dip Macroscopic were the same orders with different names during the study period.
d UA Microscopy and UA Micro were the same orders with different names during the study period.
Data collection
Patient and laboratory data were abstracted from the hospital medical informatics database. Data included patient demographics (ie, age, race, and sex), laboratory test results (ie, urinalysis, microscopic exam and urine culture), and discharge disposition (ie, home, other facility, etc). For urine cultures with accompanying urinalysis or microscopy, the time between the urine culture and urinalysis and/or microscopy was calculated. Type of urine culture specimen was also noted (ie, clean catch, catheterized, or procedure related) as indicated by the ordering clinician. For patients with multiple urine cultures during an admission, each sample was treated as an independent observation.
Definitions
Urine cultures with growth of ≥100,000 colony-forming units (CFU)/mL for a clean-catch specimen and ≥10,000 CFU/mL for a catheterized specimen were considered positive results. Urine cultures that were negative for significant growth or contaminated were considered negative for this analysis. Leukocyte esterase ≥1 identified on urinalysis and >5 white blood cells per high-power field on urine microscopy were treated as abnormal and positive test results. We defined an isolated urine culture as a culture without an associated urinalysis and/or urine microscopy performed within 1 calendar day before or after the culture was performed. Catheter-associated urinary tract infection (CAUTI) surveillance was independently conducted by the hospital infection prevention department during the study period. A CAUTI was defined according to National Healthcare Safety Network definitions23 as a UTI where an indwelling urinary catheter was in place for >2 calendar days on the date of event, with day of device placement being day 1, and an indwelling urinary catheter was in place on the date of event or the day before. If an indwelling urinary catheter was in place for >2 calendar days and then removed, the UTI criteria must have been fully met on the day of discontinuation or the next day.
Cost assessment
Unit cost of a urine culture was obtained from the Medicare Clinical Laboratory Fee Schedule using national median Medicare payment rate of $15.00 per urine culture (not adjusted to inflation).24 Total laboratory charges for urine cultures during the preintervention and postintervention periods were calculated, and cost difference was estimated.
Statistical analysis
Patient demographics and characteristics are reported on a per-admission basis. Urine cultures rates are reported per 1,000 patient days (ie, the total patient days for all patients admitted during the study period). CAUTI rates are reported per 1,000 patient days and catheter days. Demographic characteristics and urine culture data were compared for the preintervention period and the postintervention period using the Wilcoxon rank-sum test, χ2 test, or univariable logistic regression where appropriate. An interrupted time-series model was used to analyze the impact of the intervention on urine culture rates during the study period. Data were analyzed using SAS version 9.3 software (SAS Institute, Cary, NC). The Washington University Human Research Protection Office approved this study.
Results
Patient characteristics
During the study period, 18,954 patients had ≥1 urine culture ordered during their hospital stay (11,780 during the preintervention vs 7,174 during the postintervention period) (Table 2). The median age of the patients was 62 years; ~69% of patients were white and 52.3% were female. Also, ~66% of these patients were routinely discharged home and 25.5% were discharged to or transferred to other facilities. Patients in the preintervention period were slightly younger (61 years preintervention vs 62 years postintervention; P = .015), were predominantly male (48.4% vs 46.5%; P = .012), and were routinely discharged home (66.6% vs 63.9%; P < .001) compared to the postintervention period.
Table 2. Characteristics of Patients With ≥1 Urine Cultures Obtained From Inpatients
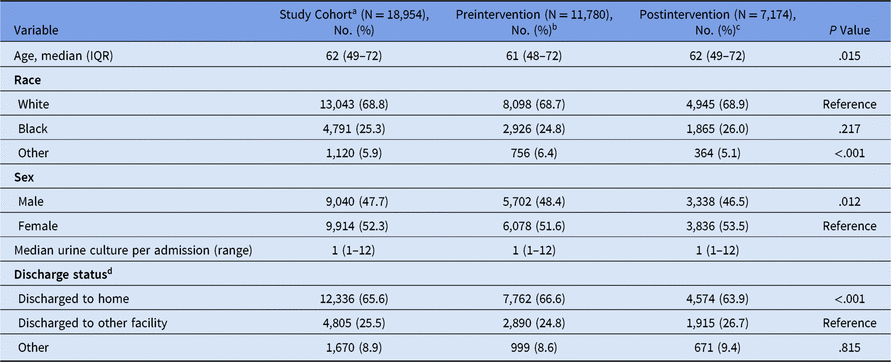
Note. IQR, interquartile range.
a Each admission is treated as an observation for this analysis purpose. Study cohort includes patients admitted to hospital whose urine was tested (≥1) for culture at the hospital during the study period (does not include urine cultures performed at the emergency department, a 24/7 clinic, or in any outpatient setting). Overall, 18,954 patient admissions contributed to 24,569 urine culture tests during the study period (median, 1; IQR, 1–1).
b January 2015 to April 2016.
c May 2016 to August 2017.
d Overall: missing (n = 143); other: still in hospital (n = 9); died (n = 1,532); left against advice (n = 106); unknown (n = 23). Preintervention: missing (n = 129); still in hospital (n = 5); admitted to hospital (n = 12); died (n = 910); left against advice (n = 72); Postintervention: missing (n = 14); still in hospital (n = 4); died (n = 622); left against advice (n = 34); unknown (n = 11).
Urine culture characteristics
A total of 24,569 urine cultures were ordered (during 18,954 admissions at the rate of 29.4 cultures per 1,000 patient days; median, 1 urine culture per admission) during the study period. Of these, 70.7% had an associated urinalysis and 70.4% had an associated microscopy (25.4% of urine cultures were deemed to be isolated). Overall, 6,642 urine cultures (27%) were positive. The proportion of positive urine cultures increased in the postintervention period (25.5% preintervention vs 29.7% postintervention; P < .001), whereas the proportion of isolated urine cultures decreased (26.0% preintervention vs 24.2% postintervention; P = .002) (Table 3).
Table 3. Comparison of Urine Culture Testing Practices Before and After the Intervention

Note: UTI, urinary tract infection; CAUTI, catheter associated urinary tract infection.
a January 2015 to April 2016.
b May 2016 to August 2017.
c See methods for definitions.
d Based upon 413,137 patient days preintervention and 421,714 patient days postintervention for all patients admitted to the hospital during the study period (see Methods).
Urine culture rates by specimen type
Urine culture decreased by 45.1% in the postintervention period (38.1 per 1,000 patient days preintervention vs 20.9 per 1,000 patient days postintervention; P < .001) (Table 3). This decrease was observed for clean catch (30.0 per 1,000 patient days preintervention vs 18.7 per 1,000 patient days postintervention; P < .001) and catheterized urine cultures (7.8 per 1,000 patient days preintervention vs 1.9 per 1,000 patient days postintervention; P < .001), whereas procedure-related urine cultures remained stable at 0.3 per 1,000 patient days (Fig. 1).

Fig. 1. Urine culture rate by specimen type. *P value for clean-catch and catheterized cultures. Note. The preintervention period was January 2015 to April 2016 and the postintervention period was May 2016 to August 2017.
When adjusted for impact of the intervention using an interrupted time series model, urine culture rates decreased significantly for overall (P < .001), catheterized (P < .001), and isolated cultures (P = .027), respectively (Fig. 2).

Fig. 2. Inpatient urine culturing practices from January 1, 2015 to August 31, 2017. The intervention time point is noted by a dashed line. P < .001 for urine cultures and catheterized cultures; P = .027 for isolated cultures, using interrupted time series analysis.
Catheter associated urinary tract infections (CAUTI)
Overall, 250 CAUTIs were identified during the study period (0.30 per 1000 patient days); however, after the intervention there was no significant change in the CAUTI rates (0.30 per 1,000 patient days preintervention vs 0.30 per 1,000 patient days postintervention; P = .871; 1.25 per 1,000 catheter days preintervention vs 1.27 per 1,000 catheter days postintervention; P = .899) (Table 3).
Effect of intervention on laboratory costs
Our intervention resulted in a $6,490 reduction in the mean monthly laboratory cost during the postintervention period, with an estimated total cost savings of $103,345 for inpatient urine culture laboratory costs in the postintervention period ($236,190 preintervention vs $132,345 in the postintervention period).
Discussion
In this retrospective study, we observed a 45.1% unadjusted decrease in the rate of inpatient urine cultures performed because of changes to electronic orders in the computer physician order entry system. The reduction in the urine culture rate was most marked for the catheterized (75.6%) compared to a clean-catch specimens (37.8%). We also noticed a 16.4% increase in the proportion of positive urine cultures and a 6.9% decrease in the proportion of isolated urine cultures obtained. Overall, our intervention resulted in an estimated reduction of $103,845 in laboratory charges to patients.
Unnecessary ordering of urine cultures and inappropriate antimicrobial use for asymptomatic bacteriuria remain common among clinicians.Reference Trautner, Grigoryan and Petersen13, Reference Nicolle, Bradley, Colgan, Rice, Schaeffer and Hooton15–17, Reference Tomford and Hershey25–Reference Forsman27 Lack of familiarity with the recommendations, excessive testing in patients with comorbidities, and certain practice patterns among physicians are some of the common factors driving this clinical practice.Reference Hartley, Valley and Kuhn9, Reference Trautner, Petersen, Hysong, Horwitz, Kelly and Naik28 Moreover, a urine culture result is often difficult for clinicians to ignore and drives antimicrobial therapy regardless of symptoms.Reference Leis, Rebick and Daneman29
Several prior efforts to prevent treatment of asymptomatic bacteriuria have included educational sessions,Reference Pavese, Saurel and Labarere6, Reference Irfan, Brooks, Mithoowani, Celetti, Main and Mertz30 pocket cards with diagnostic algorithms with audit and feedback for training clinicians,Reference Trautner, Grigoryan and Petersen13 and antimicrobial stewardship efforts. Recently, Hartley et alReference Hartley, Kuhn and Valley4 replicated these interventions in hospitalist-based service in 3 different hospitals and observed a 24% reduction in ASB treatment rates, resulting in fewer days of antimicrobial therapy. Other recent interventions have included focus groups interviews for identifying factors that affect nurse initiated urine culture ordering and collection practices,Reference Redwood, Knobloch, Pellegrini, Ziegler, Pulia and Safdar31 reflex urine culture cancellation,Reference Jones, Culbreath, Mehrotra and Gilligan21 and 2-step urine culture ordering in the emergency department.Reference Stagg, Lutz and Kirpalaney22 Although several of these upstream interventions are aimed toward eliminating unnecessary ordering and downstream interventions are aimed toward reducing treatment of asymptomatic bacteriuria, knowledge on the role of CPOE in reducing the burden of unnecessary ordering in the inpatient setting is limited.
Because of our intervention, we also noticed a significant increase in the proportion of urine cultures that were positive during the postintervention period. This finding may indicate increased clarity of reflex algorithm test names and a change in the behavior of ordering clinicians (eg, urine cultures are more likely to be ordered in patients with a higher pretest probability). The postintervention period had a significantly higher proportion of positive urine cultures with an associated abnormal or positive urinalysis (1,896 of 2,621 [72.3%] vs 2,442 of 4,021 [60.7%]; P < .001) and a significantly lower proportion of positive urine culture results with an associated negative urinalysis (122 of 2,621 [4.7%] vs 479 of 4,021 [11.9%]; P < .001). These findings suggests that a chance of an important UTI having been missed due to the decreased rate of urine culture following the intervention was less unlikely. Although we noticed a significant but small (6.9%) decrease in the isolated urine culture and substantial decrease (75.6%) in the rate of catheterized urine cultures per 1,000 patient days, there was no significant change in the CAUTI rate postintervention. Given that we previously reported that isolated urine cultures were more likely to be ordered on catheterized patients and patients with prolonged hospital stays,Reference Carlson, Munigala and Russo32 we evaluated the proportion of CAUTIs associated with isolated urine cultures. We found no significant difference between study periods in the proportion of CAUTIs that were identified based on isolated urine cultures (39 of 125 [31.2%] preintervention vs 26 of 125 [20.8%] postintervention; P = .06). These findings suggest that for patients in whom a clinical suspicion of CAUTI existed, clinicians were ordering diagnostic tests and detecting it in both intervention periods. Therefore, additional infection prevention efforts may be required in this study cohort to prevent CAUTIs.
Our intervention resulted in an estimated cost savings of ~$104,000 for inpatient laboratory costs after implementation. This represents a fraction of the total costs and does not reflect the costs saved based on the medical decisions (eg, delayed hospital discharge) and antimicrobial therapy.Reference Forsman27 In an era of reducing reimbursement for clinical laboratory testing,33 the prudent use of common diagnostic tests in patient care is increasingly important.
The limitations of our study include a retrospective design, the absence of chart review for test indication, and lack of data on antibiotic use for assessment of antimicrobial therapy. We were unable to assess asymptomatic bacteriuria because data on clinical symptoms or signs were not collected. In addition, this is a single academic medical center and may not be generalizable to other settings. Our medical informatics database does not include orders; therefore, we were unable to directly evaluate the frequency of urinalysis reflex to microscopy with culture and types of urine culture orders. We attempted to address this limitation by examining urine cultures that were performed along with urinalysis and/or microscopy, but we would not be able to identify how much our intervention reduced the proportion of urinalysis that reflexed to culture. The median number of urine cultures for the preintervention and postintervention periods were the same (including demographic characteristics patients who had >1 urine culture); therefore, we did not make any adjustments for the repeat observations. We were unable to directly assess whether antibiotic use changed in patients with urinary testing because of the intervention, and its subsequent effect on antimicrobial resistance among urinary pathogens. Strengths of our study include using data from a large academic medical center and electronic order sets for the intervention. The use of CPOE for such intervention requires relatively little ongoing intervention effort compared with other diagnostic stewardship efforts, which require constant monitoring. Our study results complement a similar CPOE intervention conducted in the emergency department of the same hospital, where we observed a 47% decrease in urine cultures ordered when only “urinalysis with reflex to microscopy” was retained in the frequently ordered list of laboratory tests.Reference Munigala, Jackups and Poirier34 A similar study of urine diagnostics reported that the elimination of reflexed microscopy examination for inpatient locations resulted in a 95% reduction in the urine microscopy performed.Reference Chen, Eintracht and MacNamara35
In summary, a staged intervention to clarify test names and inclusion of new reflex tests resulted in a 45% reduction in the urine cultures ordered with an estimated cost savings of $104,000. Further studies are needed to evaluate the role of CPOE in combination with education sessions for ordering physicians and antimicrobial stewardship efforts in reducing the incidence of unnecessary urine cultures. Future research should also focus on reducing isolated urine cultures and CAUTIs.
Author ORCIDs
David K. Warren, 0000-0001-8679-8241
Acknowledgements
None.
Financial support
This study was funded in part by Centers for Disease Control and Prevention Epicenters Program (grant no. 1U54CK000482-01).
Conflicts of interest
All authors report no conflict of interest related to this manuscript.







